- Record: found
- Abstract: found
- Article: not found
Pomc-expressing progenitors give rise to antagonistic populations in hypothalamic feeding circuits
research-article
28 March 2010
Read this article at
There is no author summary for this article yet. Authors can add summaries to their articles on ScienceOpen to make them more accessible to a non-specialist audience.
Abstract
Hypothalamic circuits regulating energy balance are highly plastic and develop in
response to nutrient and hormonal cues. To identify processes that could be susceptible
to gestational influences in the mouse, we characterized the ontogeny of proopiomelanocortin
(POMC) and neuropeptide Y (NPY) populations, which exert opposing influences on food
intake and body weight. These analyses revealed that Pomc is broadly expressed in
immature hypothalamic neurons and that half of embryonic Pomc-expressing precursors
subsequently adopt a non-POMC fate in the adult. Moreover, nearly one quarter of the
mature orexigenic NPY population shares a common progenitor with anorexigenic POMC
neurons.
The rapid increase in the prevalence of childhood obesity and the concomitant rise
in obesity-related medical morbidities and costs, lend urgency to the need for new
insights into the causes and potential preventive measures for this disease 1. Mounting
evidence supports the idea that the maternal environment can impart a lasting effect
on susceptibility of offspring to obesity and type 2 diabetes2. The arcuate nucleus
of the hypothalamus (ARH) is a critical component of the neuronal network regulating
body weight, adiposity, and glucose homeostasis; and recent studies suggest that the
development of arcuate neurons may be sensitive to maternal metabolic status 3. The
discovery that ARH projections are influenced by leptin provided the first insight
into potential mechanisms underlying “maternal programming” in the perinatal period
4. The gestational environment has also been shown to influence metabolic status of
the offspring 5; however, little is known about the embryonic origins of arcuate lineages.
The two best-characterized arcuate populations – orexigenic neurons co-expressing
neuropeptide Y (NPY) and agouti-related protein (AgRP) and anorexigenic neurons expressing
proopiomelanocortin (POMC) – produce antagonistic effects on food intake in response
to nutrient and hormonal signals of peripheral energy status (reviewed in 6). Signals
of positive energy balance, such as leptin and glucose, stimulate subsets of POMC
neurons leading to decreased food intake, while inhibiting the release of orexigenic
peptides from neighboring NPY neurons 7,8. NPY neurons are active when the energy
supply is not sufficient to meet system demands, releasing AgRP and γ-Aminobutyric
acid (GABA) to inhibit melanocortin-mediated suppression of food intake 7,9. Together,
NPY and POMC neurons integrate signals of energy homeostasis to direct physiological
processes that regulate body weight 10. We focused our initial efforts on characterizing
the ontogeny of NPY and POMC neuronal lineages during gestation because nutrient and
hormonal cues influence the formation of NPY and POMC circuits, consistent with the
idea that these developmental processes influence metabolic phenotypes.
Using the GenePaint digital mouse atlas, we found that Pomc and Npy are expressed
at embryonic day 14.5 (E14.5), whereas only Pomc is expressed at E10.5 (www.genepaint.org).
Given the earlier onset of Pomc expression, together with the established lateromedial
gradient of hypothalamic neurogenesis 11, we predicted that the lateral POMC neurons
would be born before the medial NPY neurons. To determine the birthdates of POMC and
NPY neurons in the ARH, we injected dams with a single pulse of bromodeoxyuridine
(BrdU) between E11.5–E16.5 and assessed the retention of the BrdU label by immunohistochemistry
(IHC) at postnatal day 9 (P9). Unexpectedly, analysis of BrdU label in conjunction
with Pomc or Npy expression, as assessed by fluorescent in situ hybridization (FISH)
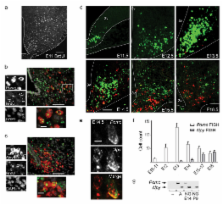
demonstrated that both populations are born between E11.5–12.5 (Fig. 1a–c). The peak
birthdate of ARH neurons is E11.5–12.5 (Fig. 1a and 12); however, we observed that
BrdU injections at E13.5 labeled some non-POMC, non-NPY cells in the lateral ARH (Supplementary
Figs. 1 and 2).
The shared birthdates of POMC and NPY neurons led us to consider whether these two
antagonistic populations of neurons may be more closely related than expected. We
characterized Pomc and Npy expression by two-color FISH across gestation (Fig. 1d–e).
Pomc expression was first observed in the hypothalamic ventricular zone at E10.5–E11.5;
from E12.5 expression was restricted to differentiated neurons, consistent with our
birthdating studies. The number of Pomc-positive (Pomc
+) cells reached a maximum at E13.5, after which its expression was extinguished in
more than half of the population between E14.5 to 18.5 (Fig. 1f). Npy expression was
not observed in the ventricular zone; it was first detected in laterally-situated
cells in the rostralmost presumptive ARH at E13.5 and subsequently expanded to more
medial and caudal regions. We did not detect appreciable levels of apoptotic cells
by TUNEL stain, consistent with the idea that Pomc expression is turned off in a large
percentage of immature hypothalamic neurons (data not shown and 13). These data argue
that Pomc expression per se does not reflect the acquisition of a terminal cell fate;
the gradual extinction of Pomc and progressive onset of Npy represent an ongoing maturation
process that extends throughout gestation. Supporting this idea, POMC and NPY neurons
do not acquire their terminal peptidergic phenotype, as reflected by Cart and Agrp
expression, until the postnatal period in rodents 14,15.
Pomc and Npy are expressed in mutually exclusive cell populations in adults 16, yet
we detected Pomc
+ and Npy
+ co-localization at mid-gestation (Fig. 1e). To substantiate the unprecedented finding
that a subset of neurons co-expresses Pomc and Npy, we compared the expression profiles
of NPY neurons isolated from embryonic versus postnatal stages. We used fluorescence
activated cell sorting (FACS) to collect GFP-positive (GFP+) cells from Npy-hrGFP
embryos, which express GFP under the control of Npy promoter and enhancer elements17.
We detected Pomc transcripts by PCR on sorted cells from E14.5, and not from P9 (Fig.
1g and Supplementary Fig. 4a,b). These observations support the idea that during gestation,
a subset of Pomc-expressing cells can differentiate into NPY neurons.
Next we used a genetic lineage tracing strategy 18 to visualize the mature POMC neuronal
population, defined by Pomc expression in adults, in relation to the broad immature
Pomc-expressing population in the embryo (Fig. 2). In Pomc-Cre;R26-GFP mice, Cre recombinase
driven by Pomc regulatory elements directs the recombination of a floxed stop codon
within the constitutively-active ROSA26 locus, permanently marking cells that expressed
Pomc from gestation 19. To assess transcriptional activity in conjunction with a GFP
reporter, we developed a technique to combine images of direct GFP fluorescence with
FISH (Supplementary Figs. 3 and 5). When this assay was performed on adult tissue
from Pomc-GFP transgenic animals, 95% of Pomc-GFP+ neurons co-express Pomc, validating
the sensitivity of this technique (Fig. 2a) 7. In contrast, only half of the GFP+
cells in Pomc-Cre;R26-GFP mice express Pomc (Figs. 2b and Supplementary Fig. 6). Pomc-negative,
GFP+ cells in Pomc-Cre;R26-GFP adults likely represent cells which turned off Pomc
expression at some point after E13.5. GFP+ cell counts in Pomc-Cre;R26-GFP animals
are consistently twice as high as those generated using Pomc FISH or direct fluorescence
in Pomc-GFP animals (Fig. 2d).
Based on our finding that Npy and Pomc are co-localized in a subset of embryonic neurons,
we considered whether some of the Pomc-negative, GFP+ neurons in Pomc-Cre;R26-GFP
adults are NPY neurons. Npy expression was detected in 17±2% of GFP+ neurons in adult
Pomc-Cre;R26-GFP mice (herafter referred to as NPYP) (Fig. 2b). We used two strategies
to independently verify this observation. First, confocal images of IHC on Npy-GFP;Pomc-Cre;R26-LacZ
mice confirmed that 25% of NPY (GFP+) neurons co-express the Pomc-Cre lineage trace
(β-Gal IHC) (Figs. 2c,e). Second, RT-PCR on FACS-purified GFP+ cells from Pomc-Cre;R26-GFP
demonstrated that some cells marked by the lineage trace express Npy (Fig. 2f and
Supplimentary Fig. 4c).
These data provide evidence that NPYP neurons are derived from progenitors that are
distinct from other ARH NPY neurons (NPYX), raising the possibility that they serve
different functions within the hypothalamic feeding circuit, and thus may underlie
the heterogeneous electrophysiological properties of NPY neurons 20. While the origins
of NPY subpopulations may differ, their subsequent differentiation converges on an
orexigenic, GABAergic phenotype, as we found that both NPYP and NPYX neurons express
Agrp and Gad67
7. Our studies provide a framework to uncover the molecular mechanism underlying these
differences within NPY neurons.
In this study, we report that Pomc is expressed in the vast majority of neurons in
the presumptive ARH (Fig. 1d,f). During gestation, Pomc transcription is extinguished
in more than half of these cells, some of which subsequently differentiate into NPY
neurons and some of which adopt alternative terminal fates. Consistent with our FISH
analyses, when Pomc-Cre;R26-GFP mice were used to trace Pomc-derived lineages in the
adult hypothalamus, we found that half of the GFP-labeled neurons are non-POMC neurons.
Therefore, use of this Pomc-Cre driver to investigate the roles played by POMC neurons
in circuits that regulate energy homeostasis would also affect a subset of NPY/AgRP
neurons and others whose functions have yet to be determined. Unanticipated effects
on antagonistic populations that also express the Cre transgene (i.e. NPY/AgRP) could
ameliorate phenotypes resulting from genetic manipulations intended for POMC neurons.
Moreover, some functions ascribed to POMC neurons could be mediated by non-POMC neurons
that also express the Cre transgene. Classification of functionally distinct subsets
of neurons derived from a Pomc
+ lineage is critical to elucidate how hormonal and nutrient signals are sensed by
ARH neurons and relayed to downtream targets that regulate body weight and energy
homeostasis.
Supplementary Material
1
2
Related collections
Most cited references16
- Record: found
- Abstract: found
- Article: not found
Rapid rewiring of arcuate nucleus feeding circuits by leptin.
Shirly Pinto, Aaron Roseberry, Hongyan Liu … (2004)
- Record: found
- Abstract: found
- Article: not found
Antagonism of central melanocortin receptors in vitro and in vivo by agouti-related protein.
Mandy L. Wilson, M Ollmann, Gregory Barsh … (1997)
- Record: found
- Abstract: found
- Article: not found
Diabetes, obesity, and the brain.
Michael W. Schwartz, Daniel Porte (2005)
