- Record: found
- Abstract: found
- Article: found
Potentiometric Sensors for Iodide and Bromide Based on Pt(II)-Porphyrin
Read this article at
Abstract
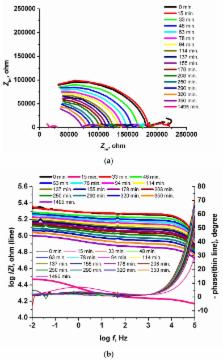
Pt(II) 5,10,15,20-tetra(4-methoxy-phenyl)-porphyrin (PtTMeOPP) was used in the construction of new ion-selective sensors. The potentiometric response characteristics (slope and selectivity) of iodide and bromide-selective electrodes based on (PtTMeOPP) metalloporphyrin in o-nitrophenyloctylether (NPOE), dioctylphtalate (DOP) and dioctylsebacate (DOS) plasticized poly(vinyl chloride) membranes are compared. The best results were obtained for the membranes plasticized with DOP and NPOE. The sensors have linear responses with near-Nernstian slopes toward bromide and iodide ions and good selectivity. The membrane plasticized with NPOE was electrochemically characterized using the EIS method to determine its water absorption and the diffusion coefficient into the membrane.
Related collections
Most cited references55
- Record: found
- Abstract: not found
- Article: not found
Potentiometric ion sensors.
- Record: found
- Abstract: found
- Article: not found
Porphyrinoids for Chemical Sensor Applications.
- Record: found
- Abstract: found
- Article: not found