- Record: found
- Abstract: found
- Article: found
Differential functional connectivity underlying asymmetric reward-related activity in human and nonhuman primates
Read this article at
Significance
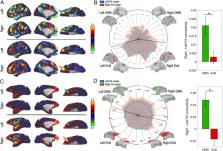
Lateralization of functions in the brain has been demonstrated in many different cognitive processes. It is supposed to increase processing abilities by reducing bilateral redundancy. Yet lateralization of reward processing, despite extremely common asymmetrical findings, has received little attention. Our neuroimaging study shows a functional lateralization of the response to reward in the lateral part of the orbitofrontal cortex (OFC), together with an asymmetric functional connectivity pattern. This particular feature was identified not only in humans but also in nonhuman primates. Our findings challenge the classical view of the OFC as a symmetrical brain region. They are urging the need for considering the specific contribution of the left and right OFC when investigating reward-related signals.
Abstract
The orbitofrontal cortex (OFC) is a key brain region involved in complex cognitive functions such as reward processing and decision making. Neuroimaging studies have reported unilateral OFC response to reward-related variables; however, those studies rarely discussed this observation. Nevertheless, some lesion studies suggest that the left and right OFC contribute differently to cognitive processes. We hypothesized that the OFC asymmetrical response to reward could reflect underlying hemispherical difference in OFC functional connectivity. Using resting-state and reward-related functional MRI data from humans and from rhesus macaques, we first identified an asymmetrical response of the lateral OFC to reward in both species. Crucially, the subregion showing the highest reward-related asymmetry (RRA) overlapped with the region showing the highest functional connectivity asymmetry (FCA). Furthermore, the two types of asymmetries were found to be significantly correlated across individuals. In both species, the right lateral OFC was more connected to the default mode network compared to the left lateral OFC. Altogether, our results suggest a functional specialization of the left and right lateral OFC in primates.
Related collections
Most cited references85
- Record: found
- Abstract: found
- Article: not found