- Record: found
- Abstract: found
- Article: not found
Longitudinal Assessment of Left Ventricular Function in Patients with Myopericarditis After mRNA COVID-19 Vaccination
Read this article at
Abstract
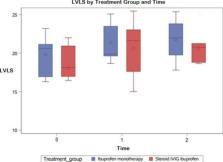
Background: Multiple reports have described myopericarditis following mRNA COVID-19 vaccination. However, data on the persistence of subclinical myocardial injury assessed by left ventricular (LV) longitudinal strain (LVLS) is limited. Objectives: Our aim was to assess LV function longitudinally in our cohort of COVID-19 vaccine-related myopericarditis using ejection fraction (EF), fractional shortening (FS), LVLS, and diastolic parameters. Methods: Retrospective, single-center review of demographic, laboratory, and management data was performed on 20 patients meeting diagnostic criteria for myopericarditis after mRNA COVID-19 vaccination. Echocardiographic images were obtained on initial presentation (time 0), at a median of 12 days (7.5, 18.5; time 1), and at a median of 44 days (29.5, 83.5; time 2). FS was calculated by M-mode, EF by 5/6 area-length methods, LVLS by utilization of TOMTEC software, and diastolic function by tissue Doppler. All parameters were compared across pairs of these time points using Wilcoxon signed-rank test. Results: Our cohort consisted predominantly of adolescent males (85%) with mild presentation of myopericarditis. The median EF was 61.6% (54.6, 68.0), 63.8% (60.7, 68.3), 61.4% (60.1, 64.6) at times 0, 1, and 2, respectively. Upon initial presentation, 47% of our cohort had LVLS < -18%. The median LVLS was -18.6% (-16.9, -21.0) at time 0, -21.2% at time 1 (-19.4, -23.5) (p = 0.004) and -20.8% (-18.7, -21.7) at time 2 (p = 0.004, as compared to time 0). Conclusions: Though many of our patients had abnormal strain during acute illness, LVLS improved longitudinally, indicating myocardial recovery. LVLS can be used as marker of subclinical myocardial injury and risk stratification in this population.
Related collections
Most cited references3
- Record: found
- Abstract: not found
- Article: not found
Myocarditis Cases Reported After mRNA-Based COVID-19 Vaccination in the US From December 2020 to August 2021
- Record: found
- Abstract: found
- Article: not found
Assessment of Prognostic Value of Left Ventricular Global Longitudinal Strain for Early Prediction of Chemotherapy-Induced Cardiotoxicity
- Record: found
- Abstract: not found
- Article: not found
