- Record: found
- Abstract: found
- Article: found
17β-Estradiol Modulates SIRT1 and Halts Oxidative Stress-Mediated Cognitive Impairment in a Male Aging Mouse Model
Read this article at
Abstract
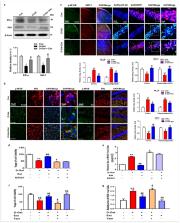
Oxidative stress has been considered the main mediator in neurodegenerative disease and in normal aging processes. Several studies have reported that the accumulation of reactive oxygen species (ROS), elevated oxidative stress, and neuroinflammation result in cellular malfunction. These conditions lead to neuronal cell death in aging-related neurodegenerative disorders such as Alzheimer’s disease (AD) and Parkinson’s disease. Chronic administration of d-galactose ( d-gal) for a period of 10 weeks causes ROS generation and neuroinflammation, ultimately leading to cognitive impairment. In this study, we evaluated the estrogen receptor α (ERα)/silent mating type information regulation 2 homolog 1 (SIRT1)-dependent antioxidant efficacy of 17β-estradiol against d-gal-induced oxidative damage-mediated cognitive dysfunction in a male mouse model. The results indicate that 17β-estradiol, by stimulating ERα/SIRT1, halts d-gal-induced oxidative stress–mediated JNK/NF-ҡB overexpression, neuroinflammation and neuronal apoptosis. Moreover, 17β-estradiol ameliorated d-gal-induced AD-like pathophysiology, synaptic dysfunction and memory impairment in adult mouse brains. Interestingly, inhibition of SIRT1 with Ex527 (a potent and selective SIRT1 inhibitor) further enhanced d-gal-induced toxicity and abolished the beneficial effect of 17β-estradiol. Most importantly, for the first time, our molecular docking study reveals that 17β-estradiol allosterically increases the expression of SIRT1 and abolishes the inhibitory potential of d-ga. In summary, we can conclude that 17β-estradiol, in an ERα/SIRT1-dependent manner, abrogates d-gal-induced oxidative stress–mediated memory impairment, neuroinflammation, and neurodegeneration in adult mice.
Related collections
Most cited references55
- Record: found
- Abstract: found
- Article: not found
Sirtuins in mammals: insights into their biological function.
- Record: found
- Abstract: not found
- Article: not found
Cloning of a novel receptor expressed in rat prostate and ovary.
- Record: found
- Abstract: found
- Article: not found