- Record: found
- Abstract: found
- Article: found
Ischemia-Triggered Glutamate Excitotoxicity From the Perspective of Glial Cells
Read this article at
Abstract
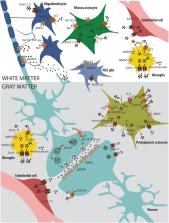
A plethora of neurological disorders shares a final common deadly pathway known as excitotoxicity. Among these disorders, ischemic injury is a prominent cause of death and disability worldwide. Brain ischemia stems from cardiac arrest or stroke, both responsible for insufficient blood supply to the brain parenchyma. Glucose and oxygen deficiency disrupts oxidative phosphorylation, which results in energy depletion and ionic imbalance, followed by cell membrane depolarization, calcium (Ca 2+) overload, and extracellular accumulation of excitatory amino acid glutamate. If tight physiological regulation fails to clear the surplus of this neurotransmitter, subsequent prolonged activation of glutamate receptors forms a vicious circle between elevated concentrations of intracellular Ca 2+ ions and aberrant glutamate release, aggravating the effect of this ischemic pathway. The activation of downstream Ca 2+-dependent enzymes has a catastrophic impact on nervous tissue leading to cell death, accompanied by the formation of free radicals, edema, and inflammation. After decades of “neuron-centric” approaches, recent research has also finally shed some light on the role of glial cells in neurological diseases. It is becoming more and more evident that neurons and glia depend on each other. Neuronal cells, astrocytes, microglia, NG2 glia, and oligodendrocytes all have their roles in what is known as glutamate excitotoxicity. However, who is the main contributor to the ischemic pathway, and who is the unsuspecting victim? In this review article, we summarize the so-far-revealed roles of cells in the central nervous system, with particular attention to glial cells in ischemia-induced glutamate excitotoxicity, its origins, and consequences.
Related collections
Most cited references308
- Record: found
- Abstract: found
- Article: not found
Cell Biology of Astrocyte-Synapse Interactions
- Record: found
- Abstract: found
- Article: not found
Aquaporin-4 deletion in mice reduces brain edema after acute water intoxication and ischemic stroke.
- Record: found
- Abstract: found
- Article: not found
Differential regulation of AMPA receptor and GABA receptor trafficking by tumor necrosis factor-alpha.
Author and article information
Comments
Comment on this article
 Smart Citations
Smart CitationsSee how this article has been cited at scite.ai
scite shows how a scientific paper has been cited by providing the context of the citation, a classification describing whether it supports, mentions, or contrasts the cited claim, and a label indicating in which section the citation was made.