- Record: found
- Abstract: found
- Article: found
Case Study: Remission of Type 2 Diabetes After Outpatient Basal Insulin Therapy
research-article
Read this article at
There is no author summary for this article yet. Authors can add summaries to their articles on ScienceOpen to make them more accessible to a non-specialist audience.
Abstract
Diabetes is a chronic, progressive disease with potentially serious sequelae. Treatment
for type 2 diabetes often begins with oral agents and eventually requires insulin
therapy. As the disease progresses, drug therapies are often intensified and rarely
reduced to control glycemia. Conversely, in type 1 diabetes, some patients experience
a “honeymoon period” shortly after diagnosis, wherein insulin needs decrease significantly
before intensification is needed (1). No comparable honeymoon period has been widely
described for type 2 diabetes. However, a few studies have demonstrated that drug-free
glycemic control can be achieved in type 2 diabetes for 12 months on average after
a 2-week continuous insulin infusion (2–4). Here, we describe an unusual case of a
26-month drug holiday induced with outpatient basal insulin in a patient newly diagnosed
with type 2 diabetes.
Case Presentation
A 69-year-old white woman (weight 72.7 kg, height 59 inches, BMI 32.3 kg/m2) was diagnosed
with type 2 diabetes in June 2011. She presented with an A1C of 17.6% (target <7%)
and a fasting blood glucose (FBG) of 452 mg/dL (target 70–130 mg/dL). Before diagnosis,
the patient had not used any oral or parenteral steroids nor had she experienced any
traumatic physical or emotional event or illness that could have abruptly increased
her blood glucose. Metformin 500 mg twice daily was initiated at diagnosis, but was
discontinued 9 days later to avoid risk of lactic acidosis, as her serum creatinine
was 1.5 mg/dL. At that time, her fasting self-monitoring of blood glucose (SMBG) values
ranged from 185 to 337 mg/dL. Treatment with 25 units of insulin detemir daily (0.34
units/kg/day) was initiated in place of metformin. The patient was counseled on diet
modifications and encouraged to exercise.
One month later (July 2011), the patient’s fasting SMBG values had improved to a range
of 71–212 mg/dL with a single hypoglycemic episode (58 mg/dL); her weight and BMI
increased slightly to 74.1 kg and 32.9 kg/m2, respectively. Hypoglycemia education
was reinforced, and insulin therapy was switched from 25 units of detemir delivered
with the Levemir FlexPen to 28 units (0.38 units/kg/day) of insulin glargine delivered
with the Lantus SoloStar due to the patient’s preference for this device. Two weeks
later, the patient reported continued improvements in fasting SMBG (70–175 mg/dL)
with one hypoglycemic episode (67 mg/dL). In response to the hypoglycemic episode,
her insulin glargine dose was decreased to 25 units daily.
In September, the patient reported fasting SMBG values ranging between 71 and 149
mg/dL, and her A1C was 7.9%. On days when the patient skipped lunch, her midday blood
glucose level would drop to <70 mg/dL (54–60 mg/dL). She was counseled not to skip
meals, and her insulin glargine dose was maintained.
In October, the patient’s weight was 71.4 kg, and her BMI was 31.7 kg/m2. She reported
recently initiating a cinnamon supplement and switching her beverage intake from sugar-sweetened
products to water and diet soda. Although the majority of her fasting SMBG values
were controlled (80–110 mg/dL), she had experienced six hypoglycemic episodes (FBG
13–64 mg/dL). All values were objectively confirmed in the patient’s glucose meter,
and the meter was replaced in case of device error. Her daily insulin glargine dose
was decreased to 20 units (0.28 units/kg/day).
In December, her SMBG values ranged between 70 and 106 mg/dL preprandially and 111
and 207 mg/dL postprandially, and she had had six additional hypoglycemic episodes
(42–66 mg/dL). The patient’s weight remained stable at 71.4 kg (BMI 31.7 kg/m2). At
this follow-up visit, her daily insulin glargine dose was decreased further to 15
units (0.21 units/kg/day).
The patient self-discontinued daily insulin glargine in March 2012 but continued using
the cinnamon supplements. She continued to perform SMBG 1–3 times/day, anticipating
loss of glycemic control. During the next 2 years, her A1C remained stable (from 6.3%
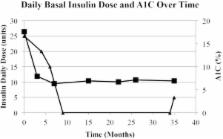
in January 2012 to 6.9% in May 2014) (Figure 1).
FIGURE 1.
Daily basal insulin dose and A1C over time. Black triangle = insulin units; black
square = A1C.
At a follow-up visit in May 2014, the patient’s SMBG indicated a need for resumed
drug therapy (FBG 107–169 mg/dL, postprandial blood glucose 108–328 mg/dL). Her weight
at this time was 65.5 kg (BMI 29.1 kg/m2). Insulin glargine was reinitiated at 5 units
daily (0.08 units/kg/day).
During the drug-free period of March 2012 to May 2014, the patient maintained her
lack of sugar-sweetened beverage consumption. However, she reported having difficulties
purchasing healthy food options because of financial constraints. In August 2013,
she was specifically encouraged to incorporate physical activity (walking) into her
daily routine. The patient’s weight during the drug-free interval declined from 70
kg in March 2012 to 65.5 kg in May 2014.
Discussion
Hyperglycemia causes pancreatic β-cell toxicity, leading to decreased insulin release
(3). In type 1 diabetes, the honeymoon period occurs when residual pancreatic β-cell
function is partially restored for an average of 7.2 months, as hyperglycemic stress
is removed before the β-cells are ultimately destroyed (1,3).
Past studies demonstrated induction of a drug-free period when patients newly diagnosed
with type 2 diabetes were treated with 2–3 weeks of intensive insulin therapy (2–5).
Ilkova et al. (2) induced a 12-month drug-free period in 46.2% (n = 6) of patients
using an insulin infusion averaging 0.61 units/kg/day. Three patients maintained glycemic
control for 37–59 months. Li et al. (3) also induced a 12-month drug-free period in
47.1% (n = 32) of patients with an insulin infusion of 0.7 units/kg. Additional studies
indicate that basal-bolus insulin therapy (0.37–0.74 units/kg/day) using NPH and regular
insulin can also induce a 12-month drug-free period in a similar percentage of patients
(43.8–44.9%) (4,5).
The mechanism of remission appears to be related to resumption of endogenous insulin
production after glucotoxicity is resolved. Glucotoxicity has been shown to inhibit
first-phase insulin secretion from the pancreatic β-cells (3). Li et al. (3) theorized
that an insulin infusion corrects hyperglycemia and removes stress from the β-cells,
allowing them to produce insulin, resulting in euglycemia. Their study quantified
an increase in secretion of endogenous insulin (44%) and C-peptide (26%) after 2 weeks
of continuous insulin infusion. The mechanism through which insulin induces a period
of drug-free glycemic control in type 2 diabetes appears to be similar to that causing
the honeymoon period in type 1 diabetes.
To our knowledge, this is the first report of basal insulin monotherapy–induced remission
of type 2 diabetes. Previous studies required multiple daily injections in a basal-bolus
therapy regimen using NPH and regular insulin or hospitalization of patients administered
a continuous insulin infusion (2–5).
Basal-only insulin therapy may be a slower method of achieving remission compared
to more intensive insulin regimens. In this case, basal insulin was maintained for
9 months. However, according to the FBG trend, discontinuation could have occurred
sooner. This report suggests that a trial of basal insulin dosed at 0.2–0.3 units/kg/day,
with follow-up every 2–4 weeks in severely hyperglycemic patients with newly diagnosed
type 2 diabetes, may be an alternative method to achieving temporary remission. Although
this insulin regimen requires a longer timeframe compared to remission induced by
basal-bolus therapy or continuous insulin infusion, it provides a more convenient
outpatient therapeutic option at a lower cost.
Limitations of this case study include the patient’s use of cinnamon supplementation,
which was continued throughout the drug-free period. Although reports are conflicting
regarding its efficacy in type 2 diabetes, it is possible that cinnamon may have exerted
a mild antidiabetic effect. Positive cinnamon studies have demonstrated a 0.36% A1C
reduction after 3 months of use (6). Additionally, the patient’s weight declined by
3.75% during the 9 months of basal insulin therapy, which was likely in response to
introducing dietary modifications related to beverage consumption. Most studies suggest
that an A1C reduction of 0.36% (7) to 0.66% (8) can be achieved with intensive lifestyle
interventions. Therefore, it is unlikely that cinnamon in combination with the mild
lifestyle modifications accounted for a nearly 11% A1C reduction from baseline.
Eliminating the consumption of sugar-rich beverages alters the postprandial glycemic
curve. In clinical practice, suppressing postprandial blood glucose excursions by
adopting significant dietary improvements may postpone or obviate the need for bolus
insulin therapy. Likewise, the remission of diabetes potentially may be achieved,
as seen in this case, with monotherapy basal insulin when dietary modifications significantly
alter the postprandial glycemic curve. However, it is unknown whether remission can
be achieved using basal insulin administration alone in patients who choose not to
incorporate lifestyle modifications or in patients with baseline healthy eating and
exercise habits.
Although weight changes did not appear to contribute to disease remission, the moderate
weight loss (6.5%) achieved during the drug-free interval and continued SMBG both
may have contributed to maintaining and extending the remission period. The Diabetes
Prevention Program (9) showed that lifestyle modifications aimed at achieving a 7%
reduction of weight significantly delay the onset of diabetes compared to placebo
and metformin. Finally, performing SMBG through the drug-free period may have empowered
the patient by providing objective criteria necessary to validate the benefits of
lifestyle modifications.
Based on this case, it is possible that initial type 2 diabetes management with basal
insulin can temporarily restore β-cell function to a degree to which blood glucose
control can be maintained without drug therapy. Although previous studies conducted
with intensive insulin regimens have reported response rates nearing 50% for ∼12 months
(2–5), future studies should investigate the ideal basal dose, percentage of patient
responders, duration of drug-free glycemic control, and mechanism through which this
phenomenon occurs. This case further highlights the need to educate every newly diagnosed
patient about the treatment of hypoglycemic events.
Summary
The purposeful remission of diabetes is not widely attempted or generally considered
possible. Although literature exists regarding the temporary honeymoon period experienced
after insulin initiation in some people with type 1 diabetes (1), comparatively little
research is available regarding the influence of insulin on the remission of type
2 diabetes. Current literature suggests benefit in nearly 50% of patients newly diagnosed
with type 2 diabetes using one of the following strategies: a 2-week inpatient insulin
infusion or multiple daily injections of basal-bolus therapy (2–5). However, there
are disadvantages to these methods. A continuous insulin infusion requires inpatient
admission, whereas a basal-bolus insulin regimen requires purchase of two products
and administration of multiple subcutaneous injections daily. Unfortunately, both
methods may be impractical, costly, and inconvenient for many patients newly diagnosed
with type 2 diabetes.
This case outlines a third potential option for inducing remission of type 2 diabetes:
basal insulin monotherapy. Using this approach avoids the costly and inconvenient
hospital admission required for the continuous insulin infusion strategy. Furthermore,
the cost of drug therapy is reduced with the purchase of one rather than two insulin
products, as needed in a basal-bolus insulin regimen. Additionally, using basal insulin
alone reduces the risk of hypoglycemic events that may occur with stacking of multiple
insulin products. Finally, requiring only one injection of insulin each day offers
a more manageable alternative for newly diagnosed patients compared to the multiple
daily injections required with a basal-bolus insulin regimen.
By using this basal insulin strategy, the patient in this case was able to achieve
drug-free glycemic control for 26 months. Early initiation of basal insulin monotherapy
in patients newly diagnosed with type 2 diabetes is a more convenient and cost-effective
approach than methods previously described and could potentially induce remission
of type 2 diabetes in other patients.
Related collections
Most cited references5
- Record: found
- Abstract: found
- Article: not found
Effect of intensive insulin therapy on beta-cell function and glycaemic control in patients with newly diagnosed type 2 diabetes: a multicentre randomised parallel-group trial.
Jianping Weng, Yanbing Li, Wen Xu … (2008)
- Record: found
- Abstract: found
- Article: not found
Induction of long-term glycemic control in newly diagnosed type 2 diabetic patients is associated with improvement of beta-cell function.
Bin Yao, Wen Xu, Yanbing Li … (2004)
- Record: found
- Abstract: found
- Article: not found
Glycated haemoglobin and blood pressure-lowering effect of cinnamon in multi-ethnic Type 2 diabetic patients in the UK: a randomized, placebo-controlled, double-blind clinical trial.
R Akilen, A Tsiami, D Devendra … (2010)