- Record: found
- Abstract: found
- Article: found
Current understanding of the role of Adipose-derived Extracellular Vesicles in Metabolic Homeostasis and Diseases: Communication from the distance between cells/tissues
Read this article at
Abstract
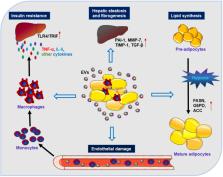
Extracellular vesicles (EVs) including exosomes, microvesicles (MVs), and apoptotic bodies, are small membrane vesicular structures that are released during cell activation, senescence, or programmed cell death, including apoptosis, necroptosis, and pyroptosis. EVs serve as novel mediators for long-distance cell-to-cell communications and can transfer various bioactive molecules, such as encapsulated cytokines and genetic information from their parental cells to distant target cells. In the context of obesity, adipocyte-derived EVs are implicated in metabolic homeostasis serving as novel adipokines. In particular, EVs released from brown adipose tissue or adipose-derived stem cells may help control the remolding of white adipose tissue towards browning and maintaining metabolic homeostasis. Interestingly, EVs may even serve as mediators for the transmission of metabolic dysfunction across generations. Also, EVs have been recognized as novel modulators in various metabolic disorders, including insulin resistance, diabetes mellitus, and non-alcoholic fatty liver disease. In this review, we summarize the latest progress from basic and translational studies regarding the novel effects of EVs on metabolic diseases. We also discuss EV-mediated cross-talk between adipose tissue and other organs/tissues that are relevant to obesity and metabolic diseases, as well as the relevant mechanisms, providing insight into the development of new therapeutic strategies in obesity and metabolic diseases.
Related collections
Most cited references92
- Record: found
- Abstract: found
- Article: not found
Sperm tsRNAs contribute to intergenerational inheritance of an acquired metabolic disorder.
- Record: found
- Abstract: found
- Article: not found
Electron microscopic evidence for externalization of the transferrin receptor in vesicular form in sheep reticulocytes
- Record: found
- Abstract: found
- Article: not found