- Record: found
- Abstract: found
- Article: found
Crystal structure of (2-amino-7-methyl-4-oxidopteridine-6-carboxylato-κ 3 O 4, N 5, O 6)aqua(1,10-phenanthroline-κ 2 N, N′)copper(II) trihydrate
Read this article at
Abstract
In a hydrated copper(II) complex, 2-amino-7-methyl-4-oxidopteridine-6-carboxylate and 1,10-phenanthroline ligands chelate the Cu II cation while a water molecule further coordinates to the Cu II cation to complete the elongated distorted octahedral coordination geometry.
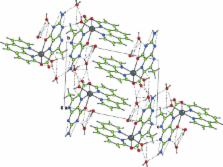
Abstract
In the title compound, [Cu(C 8H 5N 5O 3)(C 12H 8N 2)(H 2O)]·3H 2O, the Cu II cation is O, N, O′-chelated by the 2-amino-7-methyl-4-oxidopteridine-6-carboxylate anion and N, N′-chelated by the 1,10-phenanthroline (phen) ligand. A water molecule further coordinates to the Cu II cation to complete the elongated distorted octahedral coordination geometry. In the molecule, the pteridine ring system is essentially planar [maximum deviation = 0.055 (4) Å], and its mean plane is nearly perpendicular to the phen ring system [dihedral angle = 85.97 (3)°]. In the crystal, N—H⋯O, O—H⋯N and O—H⋯·O hydrogen bonds, as well as weak C—H⋯O hydrogen bonds and C—H⋯π interactions, link the complex molecules and lattice water molecules into a three-dimensional supramolecular architecture. Extensive π–π stacking between nearly parallel aromatic rings of adjacent molecules are also observed, the centroid-to-centroid distances being 3.352 (2), 3.546 (3), 3.706 (3) and 3.744 (3) Å.