- Record: found
- Abstract: found
- Article: found
Colligative Property of ATP: Implications for Enteric Purinergic Neuromuscular Neurotransmission
discussion

Read this article at
There is no author summary for this article yet. Authors can add summaries to their articles on ScienceOpen to make them more accessible to a non-specialist audience.
Abstract
ATP, a common constituent of vesicles, reduces intravesicular osmotic pressure by
polymerizing vesicular contents and reducing the number of individual free particles
A recent report by Estévez-Herrera et al. (2016) suggested that ATP, the energy coin,
also a neurotransmitter, controls the osmotic pressure of vesicular contents (Estévez-Herrera
et al., 2016). Since the discovery and description of quantal transmission (Katz,
1971; Bennett and Kearns, 2000), membrane-delimited packeted structures, the vesicles,
have been morphologically identified and correlated with secretion in several secretory
tissues, including nerve terminals of central and peripheral neurons, chromaffin cells,
platelets, and insulin-secreting beta cells of the pancreas (Goyal and Chaudhury,
2013; Südhof, 2013; Thorn et al., 2016). Though it is now known for some time that
ATP is concentrated in the vesicles, the demonstration that ATP, by virtue of being
negatively charged, and its ability to associate with positively charged molecules
like amines, agglomerates vesicular particles, is a significant finding. Vesicles
do not release their contents randomly within the cytosol, but rather are transported
to the membranes for exocytosis (Jena, 2009). An important contributor to this site-specific
release of vesicular contents may be the constant fine tuning and maintenance of osmotic
pressure isotonic with the cytosol of the vesicle-containing structure. In the light
of such plausible dynamic regulation of osmotic pressure of vesicles, the demonstration
of the likely role of ATP in regulating this vesicular osmotic pressure acquires importance
(Estévez-Herrera et al., 2016). In this perspective, we discuss the implications of
these findings on enteric purinergic inhibitory musculomotor neurotransmission.
Vesicle membrane integrity is maintained by lowering intravesicular osmotic pressure
Osmotic property is a colligative property: it depends on the number of particles,
as suggested by Raoult:
pV = nRT…standard gas equation
πV = nRT….π = osmotic pressure
π = [n/V]RT…note the dependence of π on n, the number of particles, the essence of
colligative property
The process of release of neurotransmitters is highly coordinated, involving several
100 proteins with graded responses to intracellular calcium fluctuations (Goyal and
Chaudhury, 2013; Südhof, 2013). In neurosecretory processes like stimulation-evoked
neurotransmission, the pool of readily releasable vesicles empty contents after docking
at the cell membrane of the active zones. Electron micrographs of nerve terminal varicosities
always demonstrate intact membranes of vesicles within the cytosol of the terminal
(Collman et al., 2015). This structural integrity of vesicles strongly suggests that
the contents of the vesicles are isotonic with the matrix of the varicosities.
Earlier, an interesting study investigated the osmotic pressure of synaptic vesicles
(Kopell and Westhead, 1982). This study revealed that the vesicles obtained from chromaffin
cells of the adrenal gland, despite their high concentrations of various amines, and
peptides, remained isotonic (Kopell and Westhead, 1982). This study hypothesized that
the highly negatively charged ATP molecules, a major co-constituent of chromaffin
cells beside the positively-charged amines, forms a polymeric complex within the vesicles
(Kopell and Westhead, 1982).
The leading hypothesis by Estévez-Herrera et al. (2016) is that when ATP agglomerates
the vesicular contents, there is a reduction in the number of free particles, leading
to balance of pressures across the vesicular membrane. This physical property may
be potentially a critical determinant of membrane integrity of vesicles from their
biogenesis until the time they receive the necessary stimuli for exocytosis.
SLC17A9 transports ATP into vesicles
Estévez-Herrera et al. (2016) show that SLC17A9, the vesicular nucleotide transporter
(VNUT; Sawada et al., 2008), performs a rate-limiting step to the transport of ATP
within the large dense core (LDC) particles of the chromaffin cells. It has been specifically
demonstrated earlier and well-known for some time that the drive for ATP entry is
regulated by a proton motive force (Sawada et al., 2008). Thus, it is imperative that
the colligative property has a direct relationship with the intravesicular acidity.
Enteric inhibitory smooth muscle neurotransmission involves release of vesicular ATP
and de novo synthesized nitric oxide (NO)
The biophysical characteristics of particle-based actions of ATP may have important
implications for enteric neuromuscular transmission. Evoked enteric inhibitory neuromuscular
neurotransmission involves the sequential release of purines (most importantly, ATP)
and the gas nitric oxide (NO), synthesized by neuronal nitric oxide synthase (nNOS)
at the membranes of nerve terminals (Chaudhury et al., 2011, 2012; Chaudhury, 2014,
2015a, 2016a,b). While ATP is stored in the vesicles of the nerve terminals, NO is
synthesized de novo (Chaudhury, 2016a). The released ATP during evoked neurotransmission
hyperpolarizes the smooth muscle membrane (Chaudhury, 2016a). In a span of a few 100
ms, the membrane potential endeavors to swing back to its resting stage. However,
the prolonged release of nitric oxide prevents the restoration of membrane potential
to baseline and aims to maintain the hyperpolarization. This is manifested as the
slow inhibitory junction potential (sIJP), unambiguously recorded by several investigators
across decades (Figure 1; Bennett et al., 1966; Atanasova et al., 1972; Smith et al.,
1990; Hirst et al., 2004; Allego et al., 2008; Chaudhury et al., 2011, 2012; Chaudhury,
2016a). Following the paradigm-shifting demonstration of ATP as a neurotransmitter
using gut tissues (Burnstock et al., 1970), there was a gap of several decades in
which the VNUT could not be identified within the synaptic vesicles. Quinacrine, the
antimalarial drug, robustly stains ATP containing nerve terminals (Belai and Burnstock,
1994), but this never could provide insights into how ATP, a highly negatively charged
molecule, could be shuttled across the cell membrane of the vesicles. Following the
report by Sawada et al. (2008) of the molecular identity of VNUT as the solute carrier
protein SLC17A9 (Sawada et al., 2008), a commercially available antibody was used
to demonstrate the existence of SLC17A9 in the enteric musculomotor nerve terminals
(Chaudhury et al., 2012), providing the preliminary critical evidence of fulfillment
of the Sherringtonian criterion (Levine, 2007) for the existence of the transporter
of a neurotransmitter.
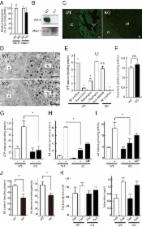
Figure 1
Colligative property of ATP may have important implications for enteric inhibitory
neuromuscular neurotransmission. (I) Trace of a compound inhibitory junction potential
Note the fast phase of the hyperpolarization (fast IJP, ATP mediated), followed by
the slow delayed phase to repolarization (slow IJP, NO mediated). ATP is released
from vesicles, whereas NO is synthesized de novo by nNOS. However, the identity of
the ATP containing vesicles is not discretely described for myenteric axons and nerve
terminals. (II) Three traces of electrical recordings showing differential responses
to electrical field stimulation (EFS) intensity The upper trace is the mechanical
recording, whereas the lower trace depicts the electrical activity. The three traces
corresponds to 1, 10, and 30 Hz of stimuli, respectively. Note that at the beginning
of the stimulus, inhibitory neurotransmission is observed, with hyperpolarization
of the membrane potential (inhibitory junction potential, IJP). The tendency to recover
to the baseline membrane potential is less with higher intensities of stimuli. The
rapid phase of IJP is due to ATP. The slow phase is due to sustained synthesis of
NO. However, the identity of the vesicles that releases ATP is not known. A notable
feature of this recording is the excitatory junction potential (EJP) at the end of
the IJP. EJPs are mainly mediated by acetylcholine. It is possible that Ach is released
with the decay of the stimulus. It is also possible that Ach is released initially,
but the overwhelming amount of ATP, through its postjunctional effects on the P2Y1
receptor, mediates an inhibitory response. Evidence also exists that the sustained
phase of the IJP may be due to a prejunctional modulation by VIP, which is also coreleased
with ATP. (III) Further examples of sequential relaxation and contraction during mechanical
recordings G1 represents a pyloric strip, whereas G4 represents an antral strip. Note
the spontaneous contractions of the antrum. In contrast, the EFS induces relaxation
of the pyloric strip, which likely contributes to pyloric patency and gastric emptying
in the organ in vivo. (IV) Mechanical relaxations are sensitive to L-NNA, and contractions
to atropine Mechanical recordings from lower esophageal sphincter. Again, note the
sequential off-contraction following an on-relaxation during the EFS (left panel).
The middle panel shows an on-contraction. Combined L-NNA-atropine still manifests
residual relaxation. (V) Enteric synaptosomal preparations show distinct vesicular
compositions of acetylcholine and VIP Note that the fraction I is composed of only
Ach, whereas the fraction II is composed of both Ach and VIP. The significance of
this complex composition is not clear, but may potentially contribute to the excitation
seen at the tail phase of an IJP. Also note that both fractions associate with Mg2+-ATPase,
which is myosin. This could be both myosin Va and myosin II. (VI) Osmotic fragility
of enteric synaptosomal vesicles Note that the Ach-VIP containing vesicles are slightly
more fragile (as tested by incubation in a hypotonic solution) in comparison to only
acetylcholine-containing vesicles, probably due to their large size. Per the recent
study of Estévez-Herrera et al. (2016), ATP may importantly contribute to the osmotic
stability of these vesicles. (VII) Cartoon depicting the potential contribution of
colligative property of ATP to enteric neurotransmission This is a simplified version
of what may actually exist in the enteric synaptosomes. The arc represents the active
zone of the junctional membrane of the enteric varicosities. Pure ATP containing vesicles
have never been detected in myenteric preparations. They either coexist with Ach,
VIP or both Ach and VIP (this third kind not shown in the cartoon). ATP, via its colligative
property, may contribute to the regulation of release kinetics of either Ach or VIP
or both, depending upon the stimulus intensity. Reproduced with permission from Chaudhury
et al. (2011); Agoston and Whittaker (1989); Anuras et al. (1974); González et al.
(2004); Burnstock (1981).
Importance of vesicular content clustering by ATP in diverse enteric synaptosomal
vesicles
ATP is widely distributed in enteric musculomotor nerve terminals. It is present in
both VIP containing large dense core vesicles, as well as acetylcholine (Ach) containing
small clear vesicles (Figure 1). While VIP plays a significant role in inhibitory
neurotransmission and smooth muscle relaxation, Ach facilitates excitatory neurotransmission
and smooth muscle contraction. Alternate relaxation and contractions of smooth muscles
at the same location are the key factors that determine transit of luminal contents
but very little is known regarding the release kinetics of VIP and Ach, and parallel
release of ATP. Below, we discuss some of the possibilities that may happen to execute
these complex release of excitatory and inhibitory neurotransmitters during enteric
nerve-smooth muscle neurotransmission. We also discuss the potential role of colligative
property of ATP in influencing these functions.
VIP containing large dense core vesicles: In the enteric nerve terminals, what potential
colligative role does ATP play? The enteric inhibitory neurotransmission is represented
electrophysiologically by the fast and slow IJP, mediated by the purine nucleotide
ATP and NO, respectively (Chaudhury et al., 2011, 2012; Chaudhury, 2016a). While ATP
is released from vesicles (Chaudhury et al., 2012), NO is synthesized by nNOS at the
nerve terminal membrane (Chaudhury et al., 2009, 2011; Chaudhury, 2014). It is only
scantily known whether other chemicals are co-released with ATP. Classical studies
by Whittaker using enteric synaptosomes has demonstrated that many neuropeptides coexist
with ATP. One of them is vasoactive intestinal polypeptide (VIP; Agoston et al., 1988;
Whittaker, 1989). It is possible that peptide VIP is coreleased with ATP during evoked
neurotransmission (Agoston and Whittaker, 1989). Though VIP may not have a direct
impact on the slow IJP, studies have shown the important role of VIP in modulation
of presynaptic calcium concentrations, thus having an effect on both exocytosis of
ATP, as well as de novo synthesis of NO (Van Geldre and Lefebvre, 2004). This may
be a reason for earlier erroneous suggestions of VIP as the enteric inhibitory neurotransmitter
(Goyal et al., 1980; Mackenzie and Burnstock, 1980). It is possible that ATP may importantly
contribute to the osmotic pressure of the VIP containing large dense core vesicles,
which are similar to the chromaffin granules. This remains to be tested.
Acetylcholine (Ach) containing small clear vesicles (SCV): An important aspect of
coexistence of ATP in the enteric nerve terminals is that with acetylcholine (Ach).
ATP, being negatively charged, can associate with the positively charged quaternary
ammonium of acetylcholine. Ach contributes to excitatory junction potentials (EJPs)
and contractile motor responses (Anuras et al., 1974). Intriguingly, cholinergic vesicles
coexist with VIP containing large dense core vesicles (Agoston et al., 1988). The
significance of this important observation is unknown. The dynamics of release of
ATP/NO and Ach is also not known (Chaudhury, 2016a). A common observation is the occurrence
of a contractile response at the end of an episode of relaxation during post-stimulus
mechanical recordings of gastrointestinal muscle strips (Figure 1; Anuras et al.,
1974). Per the previous observation, it shall imply that a given intensity of stimulus
first supports inhibitory neurotransmission, followed by the cholinergic excitatory
response. By the time the excitatory response appears, the initial stimulus would
start decaying temporally. But what prevents simultaneous release of Ach during ATP
release? It has been shown that low frequency electrical field stimulation of synaptosomes
ex vivo released ACh by < four-fold the basal release; the simultaneously detected
VIP secretion was only slightly raised above the basal level. During high frequency
stimulation (50 Hz), VIP secretion was greatly increased (to five-fold the resting
release) whereas the release of ACh increased to only 150% of the basal output (Agoston
and Whittaker, 1989). An alternate possibility is that both ATP and Ach are coreleased,
and depending on the postjunctional responses, there is an inhibitory or excitatory
response. Sometimes, say during segmentation contractions, a long stretch may simultaneously
have sustained inhibitory purinergic–nitrergic responses (Gwynne and Bornstein, 2007).
How is cholinergic responses excluded during this activity? In the light of these
perspectives, the reductionist concepts of descending inhibitory neurotransmission
and ascending excitatory neurotransmission merits critical revision. How do the circuits
toggle between an excitatory vs. inhibitory prejunctional release? This may also result
from a summative response. Though the current concepts limit us to thinking that the
postjunctional smooth muscle responses are somewhat chaotic and stochastic in nature,
there is potential stoichiometry to how nature must have designed these enteric circuits,
including specific responses to intraluminal stimuli, and responses mediated by intrinsic
primary afferent neurons (IPANs). SLC17A9 colocalizes with vesicular acetylcholine
transporter (Chaudhury et al., 2012). Again, it remains to be examined whether ATP
contributes to colligative actions with the acetylcholine containing vesicles.
Impact of colligative property of ATP on differential release of enteric excitatory
and inhibitory neurotransmitters and neuromodulators
The biophysical experiments of estimating osmotic pressure of vesicles are challenging
to perform, and more so in an in vivo context. Enteric synaptosomal preparations may
be used to examine whether the mechanisms of ATP contributing to particle stability
(Estévez-Herrera et al., 2016) is a general phenomenon seen across all vesicular structures,
for example cholinergic containing small synaptic vesicles and VIP containing large
dense core vesicles. Additionally, polymeric vesicular contents with (ATP-neurotransmitter)n
needs demonstration, likely by estimation of the polymeric masses or by surrogate
measures of vesicular acidity. The specific gravity of the clear and dense core vesicles
have been reported (Table 1). A relevant hypothesis that may be examined is whether
the ATP contents are different between exclusive Ach containing vesicles vs. Ach-VIP
containing vesicles. If so, the particle aggregating effects of ATP may differentially
regulate release of Ach and VIP during excitatory and inhibitory neurotransmission,
respectively.
Table 1
Table showing the relative specific gravity of different enteric synaptic vesicles.
Enteric Neurotransmitter
Ach (acetylcholine)
Substance P
Somatostatin
VIP (vasoactive intestinal polypeptide)
Mean density (g/ml)
1.066
1.123
1.138
1.148
Vesicle diameter (nm)
61
65
37
110
The recent study by Estévez-Herrera et al. (2016) suggest that ATP may contribute
to the osmotic stability of these vesicles. Data obtained from Agoston et al. (1985).
Pathophysiological implications of ATP colligative property for functional bowel disorders:
lessons may be learnt from SLC17A9−/− mice
Most esophagogastrointestinal motility disorders involve dysfunction of nitrergic
biosynthesis and postjunctional smooth muscle responses (Chaudhury, 2015a,b, 2016b).
Varied mechanisms of pathophysiology finally converge on the nitrergic pathways to
cause diseases like achalasia, gastroparesis, pseudo-obstruction, megacolon, and constipation.
There are virtually no disorders in which purinergic inhibitory neurotransmission
has been found as the solitary basis of the gastrointestinal motility disorder. There
are incipient suggestions that the purinergic fast IJP may be impaired, for example
in the transitional zone in Hirschsprung's disease (Jiménez et al., 2015). It is possible
that defective ATP production or vesicular shuttle may cause subtle defects in inhibitory
neuro-smooth muscle neurotransmission. Mitochondrial ATP production is defective in
diabetes (Bagkos et al., 2014). ATP gates SLC17A9 (Sawada et al., 2008). Thus, deficient
ATP production may cause SLC17A9 channelopathy. SLC17A9 knockout mice do not show
any frank gastrointestinal phenotypic abnormalities and have normal body weight (Dr.
Richard Palmiter, personal communication). In SLC17A9 knockout mice, insulin vesicular
exocytosis is accelerated (Sakamoto et al., 2014). This may result from deficient
particle aggregating action of ATP. Whether such defects also occur in enteric vesicular
release remains to be tested. We plan to undertake further studies of the enteric
synaptosomal properties during neurotransmission to test the generalizability of colligative
property of ATP and any effect of its deficiency on purinergic neuromuscular transmission.
Author contributions
AC conceptualized and drafted manuscript. VD important intellectual participation.
WM important intellectual participation, overall supervision. All authors read and
approved final version of manuscript.
Conflict of interest statement
The authors declare that the research was conducted in the absence of any commercial
or financial relationships that could be construed as a potential conflict of interest.
Related collections
Most cited references37
- Record: found
- Abstract: found
- Article: not found
Evidence that adenosine triphosphate or a related nucleotide is the transmitter substance released by non-adrenergic inhibitory nerves in the gut.
G Burnstock, D P Satchell, Ian G. Campbell … (1970)
- Record: found
- Abstract: found
- Article: found
Impairment of vesicular ATP release affects glucose metabolism and increases insulin sensitivity
Shohei Sakamoto, Takaaki Miyaji, Miki Hiasa … (2014)