- Record: found
- Abstract: found
- Article: found
Murine analogues of etanercept and of F8-IL10 inhibit the progression of collagen-induced arthritis in the mouse
Read this article at
Abstract
Introduction
Etanercept is a fusion protein consisting of the soluble portion of the p75-tumor necrosis factor receptor (TNFR) and the Fc fragment of human IgG1, which is often used for the treatment of patients with rheumatoid arthritis. F8-IL10 is a human immunocytokine based on the F8 antibody and interleukin-10, which is currently being investigated in rheumatoid arthritis with promising clinical results. We have aimed at expressing murine versions of these two fusion proteins, in order to assess their pharmaceutical performance in the collagen-induced model of rheumatoid arthritis in the mouse.
Methods
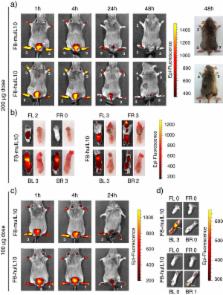
Two fusion proteins (termed muTNFR-Fc and F8-muIL10) were cloned, expressed in chinese hamster ovary (CHO) cells, purified and characterized. Biological activity of muTNFR-Fc was assessed by its ability to inhibit TNF-induced killing of mouse fibroblasts, while F8-muIL10 was characterized in terms of muIL10 activity, of binding affinity to the cognate antigen of F8, the alternatively-spliced EDA domain of fibronectin, by quantitative biodistribution analysis and in vivo imaging. The therapeutic activity of both fusion proteins was investigated in a collagen-induced mouse model of arthritis. Mouse plasma was analyzed for anti-drug antibody formation and cytokine levels were determined by bead-based multiplex technology. The association of F8-IL10 proteins with blood cells was studied in a centrifugation assay with radiolabeled protein.
Results
Both fusion proteins exhibited excellent purity and full biological activity in vitro. In addition, F8-muIL10 was able to localize on newly-formed blood vessels in vivo. When used in a murine model of arthritis, the two proteins inhibited arthritis progression. The activity of muTNFR-Fc was tested alone and in combination with F8-huIL10. The chimeric version of F8-IL10 was not better then the fully human fusion protein and showed similar generation of mouse anti-fusion protein antibodies. Incubation studies of F8-muIL10 and F8-huIL10 with blood revealed that only the fully human fusion protein is not associated with cellular components at concentrations as low as 0.2 μg/ml, thus facilitating its extravasation from blood vessels.
Related collections
Most cited references23
- Record: found
- Abstract: found
- Article: not found
Interleukin-10 therapy--review of a new approach.
- Record: found
- Abstract: found
- Article: not found
Inhibitory effect of TNF alpha antibodies on synovial cell interleukin-1 production in rheumatoid arthritis.
- Record: found
- Abstract: found
- Article: not found