- Record: found
- Abstract: found
- Article: not found
Convalescent plasma in Covid-19: Possible mechanisms of action
Read this article at
Abstract
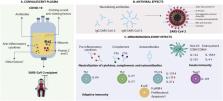
Severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) is responsible of the coronavirus disease 2019 (COVID-19) pandemic. Therapeutic options including antimalarials, antivirals, antibiotics and vaccines are under study. Meanwhile the current pandemic has called attention over old therapeutic tools to treat infectious diseases. Convalescent plasma (CP) constitutes the first option in the current situation, since it has been successfully used in other coronaviruses outbreaks. Herein, we discuss the possible mechanisms of action of CP and their repercussion in COVID-19 pathogenesis, including direct neutralization of the virus, control of an overactive immune system ( i.e., cytokine storm, Th1/Th17 ratio, complement activation) and immunomodulation of a hypercoagulable state. All these benefits of PC are expected to be better achieved if used in non-critically hospitalized patients, in the hope of reducing morbidity and mortality.
Highlights
-
•
Coronavirus disease 19 (COVID-19) is an emerging viral threat with major repercussions for public health.
-
•
There is not specific treatment for COVID-19.
-
•
Convalescent plasma (CP) emerges as the first option of management for hospitalized patients with COVID-19.
-
•
Transference of neutralizing antibodies helps to control COVID-19 infection and modulates inflammatory response.
-
•
Other plasma components may enhance the antiviral and anti-inflammatory properties of CP.
Related collections
Most cited references85
- Record: found
- Abstract: found
- Article: not found
Clinical features of patients infected with 2019 novel coronavirus in Wuhan, China
- Record: found
- Abstract: found
- Article: not found
A Novel Coronavirus from Patients with Pneumonia in China, 2019
- Record: found
- Abstract: found
- Article: not found
