- Record: found
- Abstract: found
- Article: found
Neurons, Glia, Extracellular Matrix and Neurovascular Unit: A Systems Biology Approach to the Complexity of Synaptic Plasticity in Health and Disease
Read this article at
Abstract
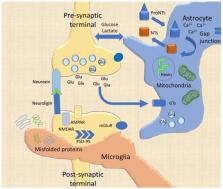
The synaptic cleft has been vastly investigated in the last decades, leading to a novel and fascinating model of the functional and structural modifications linked to synaptic transmission and brain processing. The classic neurocentric model encompassing the neuronal pre- and post-synaptic terminals partly explains the fine-tuned plastic modifications under both pathological and physiological circumstances. Recent experimental evidence has incontrovertibly added oligodendrocytes, astrocytes, and microglia as pivotal elements for synapse formation and remodeling (tripartite synapse) in both the developing and adult brain. Moreover, synaptic plasticity and its pathological counterpart (maladaptive plasticity) have shown a deep connection with other molecular elements of the extracellular matrix (ECM), once considered as a mere extracellular structural scaffold altogether with the cellular glue (i.e., glia). The ECM adds another level of complexity to the modern model of the synapse, particularly, for the long-term plasticity and circuit maintenance. This model, called tetrapartite synapse, can be further implemented by including the neurovascular unit (NVU) and the immune system. Although they were considered so far as tightly separated from the central nervous system (CNS) plasticity, at least in physiological conditions, recent evidence endorsed these elements as structural and paramount actors in synaptic plasticity. This scenario is, as far as speculations and evidence have shown, a consistent model for both adaptive and maladaptive plasticity. However, a comprehensive understanding of brain processes and circuitry complexity is still lacking. Here we propose that a better interpretation of the CNS complexity can be granted by a systems biology approach through the construction of predictive molecular models that enable to enlighten the regulatory logic of the complex molecular networks underlying brain function in health and disease, thus opening the way to more effective treatments.
Related collections
Most cited references135
- Record: found
- Abstract: found
- Article: not found
Uniquely hominid features of adult human astrocytes.
- Record: found
- Abstract: found
- Article: not found
Tripartite synapses: glia, the unacknowledged partner.
- Record: found
- Abstract: found
- Article: not found