- Record: found
- Abstract: found
- Article: found
Personalizing potassium management in patients on haemodialysis
editorial
22 October 2020
Read this article at
There is no author summary for this article yet. Authors can add summaries to their articles on ScienceOpen to make them more accessible to a non-specialist audience.
Abstract
The regulation of potassium homoeostasis changes dramatically in patients with kidney
failure who are treated by haemodialysis. With the kidney largely out of the equation,
haemodialysis patients rely on potassium removal during each dialysis session to prevent
hyperkalaemia. In addition, the gut becomes an important accessory organ for potassium
excretion [1]. Despite these adaptations, hyperkalaemia (defined as serum potassium
>5.5 mmol/L) remains a common electrolyte disorder occurring in approximately 14%
of haemodialysis patients [2]. Although seemingly counterintuitive, a minority of
haemodialysis patients is hypokalaemic (serum potassium <3.5 mmol/L), and this is
usually related to poor dietary intake [3]. The target serum potassium in haemodialysis
is unknown, but one study suggests that a serum potassium between 4.6 and 5.3 mmol/L
is associated with the greatest survival [3]. Of note, in patients with chronic kidney
disease (CKD), this optimal serum potassium range seems to be lower (4.0–4.5 mmol/L)
[4]. Furthermore , when analysing serum potassium in haemodialysis patients, it is
important to factor in when it was measured (after the long or short interdialytic
interval, time of day and seasonality), and to analyse serial measurements to exclude
transient hyperkalemia [2, 5]. Nephrologists can manage potassium balance in haemodialysis
patients in three ways, including (i) by modifying the dialysate potassium concentration,
(ii) by prescribing potassium binders and (iii) by modifying dietary potassium intake
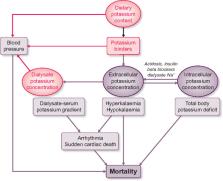
(Figure 1). The reason to implement such interventions is usually driven by recurring
predialysis hyperkalaemia and the related risk of cardiac arrhythmia [6]. Although
the prevention of acute complications is important, another relevant question is how
these interventions affect long-term outcomes in haemodialysis patients. Unfortunately,
there is a scarcity of randomized controlled trials in this area. Therefore, instead,
we need to rely on registries, which often provide useful insights into how real-world
management influences long-term outcomes. A good example of such a registry is the
French Renal Epidemiology and Information Network (REIN). In this issue, Mercadal
et al. [7] use this registry to analyse the effect of prescription patterns of dialysate
potassium and potassium binders on survival in over 25 000 patients who started haemodialysis
in 2010–13 and were followed until the end of 2014. Using Cox proportional hazard
models, the investigators show that dialysis centres that used two or three dialysate
potassium concentrations had a lower mortality risk than centres that only used one
formula. In addition, patients who used the potassium binder sodium or calcium polystyrene
sulphonate in a dose of 4–8 or ≥8 g/day had a lower mortality risk than patients who
did not use potassium binders. Conversely, patients who used potassium binders in
a dose <4 g/day had a higher mortality risk. Oral potassium supplements, which were
used in 6% of patients, were not associated with a survival benefit. What does this
study teach us on potassium management in haemodialysis patients and what are the
implications for clinical practice?
FIGURE 1
Schematic of potassium balance during haemodialysis and how it may affect mortality.
The three possibilities for intervention—dietary potassium content, potassium binders
and dialysate potassium—are shown in pink font. See text for further details.
DIALYSATE POTASSIUM
An unresolved issue is whether predialysis hyperkalaemia or a high dialysate–serum
potassium gradient is the most important risk factor for adverse outcomes in haemodialysis
patients (Figure 1) [5, 8]. The most commonly used dialysate potassium concentration
varies widely per country with some countries using predominantly 1–2 mmol/L and other
countries using 3–4 mmol/L [9]. Pun and Middleton [10] reviewed the nine retrospective
studies that analysed the association between the dialysate potassium concentration
and outcome, which usually was sudden cardiac death or all-cause mortality. Although
five studies identified an increased risk of adverse outcomes with lower dialysate
potassium (defined as <2 or <3 mmol/L), two studies found no association, and two
studies found associations that favoured lower dialysate potassium [10]. Using sales
data, Mercadal et al. [7] show that over time a lower dialysate potassium concentration
was being used less frequently. For example, the percentage of centres using <2 mmol/L
decreased from 57% to 49%, whereas the use of 3 mmol/L or ≥4 mmol/L increased from
89% to 91% and from 3% to 13%, respectively. This trend seems to echo the recent literature
in which most studies linked a low dialysate potassium to an increased risk of adverse
outcomes [10]. However, this was not a universal finding, because Mercadal et al.
[7] also showed that the occasional use of dialysate potassium <2 mmol/L was associated
with lower mortality compared with no use. This heterogeneity seems to suggest that
it is impossible to select one ideal dialysate potassium concentration, and that the
dialysate potassium prescription needs to be personalized, as proposed by others previously
[11]. The data by Mercadal et al. [7] confirm this impression, because centres that
used more dialysate potassium concentrations had lower mortality rates. A limitation
of the study by Mercadal et al. was that serum potassium measurements were not included,
and that therefore analysis of the dialysate–serum potassium gradient was not possible.
That aside, Mercadal et al. and others have called to change the practice of ‘the
regrettable routine use of a single dialysate potassium concentration’ [3, 7]. Equally
regrettable is the fact that the evidence for a positive effect of a more personalized
approach has not yet reached clinical practice. Redaelli et al. [12] performed a randomized
cross-over trial to compare a fixed dialysate potassium concentration with a strategy
during which the dialysate potassium concentration was adjusted to obtain a constant
dialysate–serum potassium gradient. The latter approach reduced the arrhythmogenic
effect of a fixed dialysate potassium concentration. It is important to emphasize
that not only low dialysate potassium but also low calcium and magnesium contribute
to this arrhythmogenic effect [10, 13]. In addition to the arrhythmogenic effect,
a lower dialysate potassium concentration may also affect blood pressure during and
after dialysis (Figure 1). Gabutti et al. [14] showed that the risk of intra-dialysis
hypotension was inversely correlated to the potassium concentration in the dialysate.
Conversely, dialysate potassium concentrations of 1 and 2 mmol/L produce ‘rebound
hypertension’ 1 h after dialysis, a phenomenon that was not observed with a dialysate
potassium concentration of 3 mmol/L [15]. In summary, the inclination to lower the
dialysate potassium concentration in haemodialysis patients with recurring hyperkalaemia
may in itself be harmful by imposing a higher dialysate–serum potassium gradient.
In this era of artificial intelligence, it must be feasible to design more individualized
dialysate potassium prescriptions that receive feedback from serial measurements of
serum potassium. Indeed, mathematical modelling of potassium profiling has been proposed
[16] and may benefit from emerging technologies using in-line monitoring of potassium
with optical ion-selective microsensors [17]. Because less potassium is removed with
a more constant dialysate–serum potassium gradient, this also implies that potassium
management in haemodialysis patients should not solely rely on the dialysate potassium
concentration.
POTASSIUM BINDERS
Potassium binders reduce serum potassium because they exchange potassium for sodium
or calcium in the gastrointestinal tract and thereby limit potassium absorption (Figure 1).
In the study by Mercadal et al. [7], 37% of patients used potassium binders at the
start of the observation period, although this decreased over time. Another French
registry study observed a much higher potassium binder prescription rate of 61% [2].
Of note, prescription may differ from actual use, as some potassium binders are poorly
palatable thereby reducing adherence. In both French registries, patients were usually
prescribed the potassium binder sodium polystyrene sulphonate [2, 7]. In a small randomized
clinical trial in patients with CKD, sodium polystyrene sulphonate was superior to
placebo and lowered serum potassium by approximately 1 mmol/L [18]. A concern regarding
the use of sodium polystyrene sulphonate, however, is that it can cause colonic necrosis
as a rare side-effect [19]. The gastrointestinal side-effects of sodium polystyrene
sulphonate were recently studied more systematically in a population-based study from
Canada and a CKD-based study from Sweden that also included patients treated with
haemodialysis [20, 21]. Both studies showed that the use of sodium polystyrene sulphonate
was associated with a higher risk of gastrointestinal complications, including intestinal
ischaemia or thrombosis, ulceration or perforation, and resection or ostomy [20, 21].
Although the incidence rate for these serious complications was still low, both studies
provided a clear signal for caution [20, 21]. In the previous 5 years, two novel potassium
binders have been introduced in nephrology and cardiology, including patiromer and
sodium zirconium cyclosilicate. Both potassium binders have recently also been studied
in haemodialysis patients. A retrospective cohort study showed that patiromer effectively
reduced serum potassium in haemodialysis patients, with an average decrease in serum
potassium of 0.5 mmol/L [22]. In a randomized, double-blind and placebo-controlled
clinical trial, sodium zirconium cyclosilicate reduced the incidence of predialysis
hyperkalaemia [23]. More specifically, 41% of patients had a predialysis serum potassium
of 4.0–5.0 mmol/L during at least three of four haemodialysis treatments after the
long interdialytic interval [23]. Although the use of patiromer and sodium zirconium
cyclosilicate appears to be safe in clinical trial settings, post-marketing surveillance
should monitor for gastrointestinal side-effects. Another consideration for all potassium
binders is that they will increase absorption of the electrolyte for which potassium
is exchanged (sodium or calcium), which could potentially contribute to sodium overload
or vascular calcification [24]. Thus, both the older and newer potassium binders may
help to manage hyperkalaemia in haemodialysis patients, but the question is how this
affects long-term outcomes. A paradoxical finding in the study by Mercadal et al.
[7] was that prescription of higher doses of potassium binders was associated with
lower mortality, whereas lower dosing was associated with higher mortality. The authors
acknowledge that these associations are likely explained by factors other than potassium
control. An important alternative explanation could be that patients receiving higher
doses of potassium binders more often consume a potassium-rich diet, which in itself
is associated with better survival [25]. Another interesting observation by another
study was that patiromer reduced blood pressure in patients with CKD, hyperkalaemia
and the use of renin–angiotensin system inhibitors [26]. Although this study requires
confirmation, an antihypertensive effect of potassium binders might be explained by
lowering of plasma aldosterone [26]. If this is a dose-dependent class effect of potassium
binders, this could also add to the explanation of why higher doses of potassium binders
associate with lower mortality (Figure 1).
DIETARY POTASSIUM INTAKE
Rather than binding potassium after dietary intake, a more direct strategy against
hyperkalaemia could be to prescribe a low potassium diet (Figure 1). In patients with
a tendency to develop hyperkalaemia, a dietary potassium intake of <3 g/day (<77 mmol/day)
is recommended [27]. Of note, the general population and patients with CKD already
consume a relatively low potassium diet [25, 28, 29]. A recent systematic review and
meta-analysis compared the effects of lower and higher dietary potassium intake in
patients with CKD (including patients treated with dialysis) on serum potassium and
mortality [30]. This review found that a potassium-restricted diet (1295 mg/day) lowered
serum potassium by 0.22 mmol/L compared with an unrestricted—but still low potassium—diet
(1570 mg/day). Furthermore, a low potassium diet (1670 mg/day) was associated with
a 40% reduction in mortality hazard compared with higher dietary potassium intake
(4414 mg/day). However, the evidence to support these effects was classified as very
low-quality evidence [30]. A small randomized controlled trial analysed the effect
of dietary potassium restriction for 2 years on nerve function in patients with CKD
Stage G3 or G4 [31]. The intervention caused modest but significant reductions in
dietary potassium intake (3272 versus 3874 g/day) and serum potassium (4.6 versus
4.8 mmol/L), which was sufficient to improve the total neuropathy score (the primary
outcome). This study illustrates that dietary potassium restriction may have benefits
beyond lowering serum potassium, although the study had several limitations [31].
At the same time, emerging evidence indicates that higher dietary potassium intake
may be beneficial, even for patients with CKD [28]. To date, 11 cohort studies analysed
the association between urinary potassium excretion (as a proxy for dietary intake)
and kidney outcomes in patients with CKD [32]. Although the majority of these studies
showed that a higher urinary potassium excretion was associated with a lower risk
of adverse kidney outcomes or mortality, this was not a consistent finding. To address
the possibility of a causal link between dietary potassium depletion and kidney outcomes,
we are currently conducting a randomized, double-blind and placebo-controlled trial
with potassium supplementation in patients with CKD [29]. Higher dietary potassium
intake has been shown to reduce blood pressure and the risk of stroke in subjects
without CKD and prevents kidney damage in experimental models of CKD [28]. Because
a high potassium diet usually consists of fruits and vegetables, potassium-independent
factors such as low animal protein, high-fiber content and an alkaline diet may also
play a role. Indeed, a randomized clinical trial showed that both alkali treatment
and fruits and vegetables can reduce kidney injury in patients with CKD [33]. To take
advantage of these effects, the need for clinical trials comparing a potassium-restricted
diet with the combination of a potassium-liberalized diet and potassium binders in
patients treated with haemodialysis has recently been emphasized [34].
TOTAL BODY POTASSIUM
This editorial so far has focused on the effects of potassium interventions on the
predialysis serum potassium concentration and the dialysate–serum potassium gradient.
However, potassium is primarily an intracellular cation, where it plays a crucial
role in cellular function. Therefore, it is also important to consider how interventions
change intracellular potassium concentration and total body potassium, and how this
relates to outcomes (Figure 1). Measurement of intracellular electrolyte concentrations
and whole-body counting of naturally radioactive potassium can be used to provide
insight into the ‘black box’ of the intracellular compartment. Unexpectedly, such
measurements reveal that patients with CKD or patients on haemodialysis often have
a total body potassium deficit, despite their tendency to develop hyperkalaemia (Figure 2)
[35, 36]. For example, the intracellular potassium concentration and the resting membrane
potential were lower in muscle cells of patients with CKD compared with healthy subjects
[37]. The initiation of haemodialysis normalized intracellular potassium, but did
not improve the resting membrane potential. Similarly, total body potassium is up
to 10% lower in haemodialysis patients compared with control subjects [35, 36] (Figure 2).
When patients transit from predialysis care to haemodialysis, total body potassium
was restored in some patients, whereas in others it decreased [38]. Preliminary data
suggest that total body potassium depletion is also associated with increased mortality
(Figure 1) [35]. Because more than 60% of potassium removal derives from the intracellular
compartment [10], dialysis may contribute to reducing total body potassium. In this
regard, the combination of a low potassium diet and a low dialysate potassium concentration
may exhaust intracellular potassium stores. Of note, hyperkalaemia and total body
potassium deficiency may co-exist if factors prevent the entry of potassium into cells.
Such factors are not uncommon in haemodialysis patients and include metabolic acidosis,
insulin resistance and the use of beta-blockers (Figure 1). Of interest, a high dialysate
sodium also contributes to the interdialytic increase in serum potassium because hypertonicity
causes a shift of potassium out of cells [39]. Conversely, one could postulate that
a higher dialysate potassium may improve sodium removal during haemodialysis and contributes
to better blood pressure control. To this end, we are currently conducting a cross-over
study to compare the effects of a dialysate potassium of 4.0 mmol/L with 2.0 mmol/L
on blood pressure, volume and intracellular sodium and potassium.
FIGURE 2
Comparison of total body potassium (TBK, ratio of measured versus expected normal)
between haemodialysis patients and controls (patients with essential hypertension).
Data are based on Williams et al. [36]. Measured TBK in controls and haemodialysis
patients were 3390 and 2810 mmol, respectively. Time on dialysis varied from <1 to
>5 years. TBK was measured before dialysis. Values are mean ± standard error.
In conclusion, to improve potassium management and potentially long-term outcomes
in haemodialysis, we believe it is important to integrate all factors that determine
potassium balance and apply a personalized approach that is dynamic and relies on
more frequent serum potassium measurements and ideally also on total body potassium.
FUNDING
The authors are supported by a consortium grant from the Dutch Kidney Foundation (CP16.01).
CONFLICT OF INTEREST STATEMENT
None declared.
(See related article by Mercadal et al. Prescription patterns of dialysate potassium
and potassium binders and survival on haemodialysis—the French Renal Epidemiology
and Information Network registry. Nephrol Dial Transplant 2021; 36: 151--159)
Related collections
Most cited references39
- Record: found
- Abstract: found
- Article: not found
Urinary sodium and potassium excretion, mortality, and cardiovascular events.
- Record: found
- Abstract: found
- Article: not found
A comparison of treating metabolic acidosis in CKD stage 4 hypertensive kidney disease with fruits and vegetables or sodium bicarbonate.
Myeong Chan Jo, Nimrit Goraya, Jan Simoni … (2013)