- Record: found
- Abstract: found
- Article: found
Nanotechnology-based strategies for treatment of ocular disease

Read this article at
Abstract
Ocular diseases include various anterior and posterior segment diseases. Due to the unique anatomy and physiology of the eye, efficient ocular drug delivery is a great challenge to researchers and pharmacologists. Although there are conventional noninvasive and invasive treatments, such as eye drops, injections and implants, the current treatments either suffer from low bioavailability or severe adverse ocular effects. Alternatively, the emerging nanoscience and nanotechnology are playing an important role in the development of novel strategies for ocular disease therapy. Various active molecules have been designed to associate with nanocarriers to overcome ocular barriers and intimately interact with specific ocular tissues. In this review, we highlight the recent attempts of nanotechnology-based systems for imaging and treating ocular diseases, such as corneal d iseases, glaucoma, retina diseases, and choroid diseases. Although additional work remains, the progress described herein may pave the way to new, highly effective and important ocular nanomedicines.
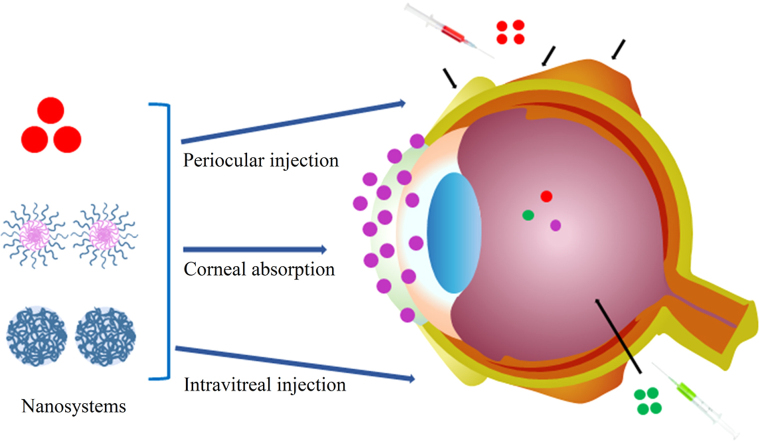
Graphical abstract
In this review, we highlights recent advances in development of nanotechnology-based systems, which could deliver both ocular drugs and gene to the eye via cornal absorption, periocular injection, and intravitreal injection, for ocular disease therapy and diagnosis. Both of nanosystems application and challenge in ophthalmology have been discussed and prospected.
Related collections
Most cited references92
- Record: found
- Abstract: found
- Article: not found
Preservatives in eyedrops: the good, the bad and the ugly.
- Record: found
- Abstract: found
- Article: not found
Challenges and obstacles of ocular pharmacokinetics and drug delivery.
- Record: found
- Abstract: found
- Article: not found