- Record: found
- Abstract: found
- Article: found
Association of genes in hereditary metabolic diseases with diagnosis, prognosis, and treatment outcomes in gastric cancer
Read this article at
Abstract
Background
Aberrant metabolism is a major hallmark of cancers and hereditary diseases. Genes associated with inborn metabolic errors may also play roles in cancer development. This study evaluated the overall impact of these genes on gastric cancer (GC).
Methods
In total, 162 genes involved in 203 hereditary metabolic diseases were identified in the Human Phenotype Ontology database. Clinical and multi-omic data were acquired from the GC cohort of the Affiliated Hospital of Jiangsu University and other published cohorts. A 4-gene and 32-gene signature was established for diagnosis and prognosis or therapeutic prediction, respectively, and corresponding abnormal metabolism scores (AMscores) were calculated.
Results
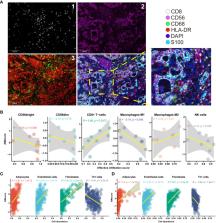
The diagnostic AMscore showed high sensitivity (0.88-1.00) and specificity (0.89-1.00) to distinguish between GC and paired normal tissues, with area under the receiver operating characteristic curve (AUC) ranging from 0.911 to 1.000 in four GC cohorts. The prognostic or predictive AMscore was an independent predictor of overall survival (OS) in five GC cohorts and a predictor of the OS and disease-free survival benefit of postoperative chemotherapy or chemoradiotherapy in one GC cohort with such data. The AMscore adversely impacts immune biomarkers, including tumor mutation burden, tumor neoantigen burden, microsatellite instability, programmed death-ligand 1 protein expression, tumor microenvironment score, T cell receptor clonality, and immune cell infiltration detected by multiplex immunofluorescence staining. The AUC of the AMscore for predicting immunotherapy response ranging from 0.780 to 0.964 in four cohorts involving GC, urothelial cancer, melanoma, and lung cancer. The objective response rates in the low and high AMscore subgroups were 78.6% and 3.2%, 40.4% and 7%, 52.6% and 0%, and 72.7% and 0%, respectively (all p<0.001). In cohorts with survival data, a high AMscore was hazardous for OS or progression-free survival, with hazard ratios ranged from 5.79 to 108.59 (all p<0.001). Importantly, the AMscore significantly improved the prediction of current immune biomarkers for both response and survival, thus redefining the advantaged and disadvantaged immunotherapy populations.
Related collections
Most cited references53
- Record: found
- Abstract: found
- Article: not found
Global cancer statistics 2020: GLOBOCAN estimates of incidence and mortality worldwide for 36 cancers in 185 countries
- Record: found
- Abstract: found
- Article: not found
TGF-β attenuates tumour response to PD-L1 blockade by contributing to exclusion of T cells
- Record: found
- Abstract: found
- Article: not found