- Record: found
- Abstract: found
- Article: found
Direct Retino-Raphe Projection Alters Serotonergic Tone and Affective Behavior
Read this article at
Abstract
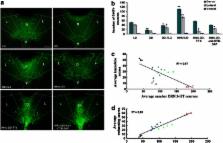
Light is a powerful modulator of higher-order cognitive processes such as mood but it remains unclear which neural circuits mediate the impact of light on affective behavior. We found that light deprivation produces a depressive-like behavioral state that is reversed by activation of direct retinal signals to the serotonergic dorsal raphe nucleus (DRN) in a manner equivalent to treatment with the selective serotonin reuptake inhibitor fluoxetine. Surprisingly, the DRN-projecting retinal ganglion cells (RGCs) are indistinguishable from the classic alpha/Y-like RGC type that contributes to image-forming visual pathways. Silencing RGC firing or specific immunotoxin ablation of DRN-projecting RGCs increased depressive-like behavior and reduced serotonin levels in the DRN. Serotonin has a key role in the pathophysiology of depression, and these results demonstrate that retino-raphe signals modulate DRN serotonergic tone and affective behavior.
Related collections
Most cited references45
- Record: found
- Abstract: found
- Article: not found
The contrast sensitivity of retinal ganglion cells of the cat.
- Record: found
- Abstract: found
- Article: not found
Assessing substrates underlying the behavioral effects of antidepressants using the modified rat forced swimming test.
- Record: found
- Abstract: found
- Article: not found
