- Record: found
- Abstract: found
- Article: found
KAF156 Is an Antimalarial Clinical Candidate with Potential for Use in Prophylaxis, Treatment, and Prevention of Disease Transmission
Read this article at
Abstract
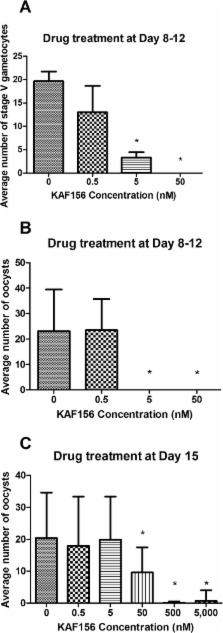
Renewed global efforts toward malaria eradication have highlighted the need for novel antimalarial agents with activity against multiple stages of the parasite life cycle. We have previously reported the discovery of a novel class of antimalarial compounds in the imidazolopiperazine series that have activity in the prevention and treatment of blood stage infection in a mouse model of malaria. Consistent with the previously reported activity profile of this series, the clinical candidate KAF156 shows blood schizonticidal activity with 50% inhibitory concentrations of 6 to 17.4 nM against P. falciparum drug-sensitive and drug-resistant strains, as well as potent therapeutic activity in a mouse models of malaria with 50, 90, and 99% effective doses of 0.6, 0.9, and 1.4 mg/kg, respectively. When administered prophylactically in a sporozoite challenge mouse model, KAF156 is completely protective as a single oral dose of 10 mg/kg. Finally, KAF156 displays potent Plasmodium transmission blocking activities both in vitro and in vivo. Collectively, our data suggest that KAF156, currently under evaluation in clinical trials, has the potential to treat, prevent, and block the transmission of malaria.
Related collections
Most cited references36
- Record: found
- Abstract: found
- Article: not found
Human malaria parasites in continuous culture.
- Record: found
- Abstract: found
- Article: not found
Drugs for bad bugs: confronting the challenges of antibacterial discovery.
- Record: found
- Abstract: found
- Article: not found