- Record: found
- Abstract: found
- Article: found
CD45 regulates retention, motility, and numbers of hematopoietic progenitors, and affects osteoclast remodeling of metaphyseal trabecules
Read this article at
Abstract
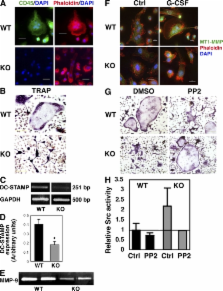
The CD45 phosphatase is uniquely expressed by all leukocytes, but its role in regulating hematopoietic progenitors is poorly understood. We show that enhanced CD45 expression on bone marrow (BM) leukocytes correlates with increased cell motility in response to stress signals. Moreover, immature CD45 knockout (KO) cells showed defective motility, including reduced homing (both steady state and in response to stromal-derived factor 1) and reduced granulocyte colony-stimulating factor mobilization. These defects were associated with increased cell adhesion mediated by reduced matrix metalloproteinase 9 secretion and imbalanced Src kinase activity. Poor mobilization of CD45KO progenitors by the receptor activator of nuclear factor κB ligand, and impaired modulation of the endosteal components osteopontin and stem cell factor, suggested defective osteoclast function. Indeed, CD45KO osteoclasts exhibited impaired bone remodeling and abnormal morphology, which we attributed to defective cell fusion and Src function. This led to irregular distribution of metaphyseal bone trabecules, a region enriched with stem cell niches. Consequently, CD45KO mice had less primitive cells in the BM and increased numbers of these cells in the spleen, yet with reduced homing and repopulation potential. Uncoupling environmental and intrinsic defects in chimeric mice, we demonstrated that CD45 regulates progenitor movement and retention by influencing both the hematopoietic and nonhematopoietic compartments.
Related collections
Most cited references42
- Record: found
- Abstract: found
- Article: not found
Recruitment of stem and progenitor cells from the bone marrow niche requires MMP-9 mediated release of kit-ligand.
- Record: found
- Abstract: found
- Article: not found
G-CSF induces stem cell mobilization by decreasing bone marrow SDF-1 and up-regulating CXCR4.
- Record: found
- Abstract: found
- Article: not found