- Record: found
- Abstract: found
- Article: found
microRNA-455-5p alleviates neuroinflammation in cerebral ischemia/reperfusion injury

Read this article at
Abstract
Abstract
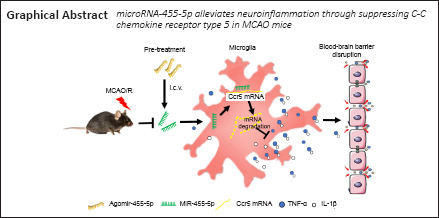
Neuroinflammation is a major pathophysiological factor that results in the development of brain injury after cerebral ischemia/reperfusion. Downregulation of microRNA (miR)-455-5p after ischemic stroke has been considered a potential biomarker and therapeutic target for neuronal injury after ischemia. However, the role of miR-455-5p in the post-ischemia/reperfusion inflammatory response and the underlying mechanism have not been evaluated. In this study, mouse models of cerebral ischemia/reperfusion injury were established by transient occlusion of the middle cerebral artery for 1 hour followed by reperfusion. Agomir-455-5p, antagomir-455-5p, and their negative controls were injected intracerebroventricularly 2 hours before or 0 and 1 hour after middle cerebral artery occlusion (MCAO). The results showed that cerebral ischemia/reperfusion decreased miR-455-5p expression in the brain tissue and the peripheral blood. Agomir-455-5p pretreatment increased miR-455-5p expression in the brain tissue, reduced the cerebral infarct volume, and improved neurological function. Furthermore, primary cultured microglia were exposed to oxygen-glucose deprivation for 3 hours followed by 21 hours of reoxygenation to mimic cerebral ischemia/reperfusion. miR-455-5p reduced C-C chemokine receptor type 5 mRNA and protein levels, inhibited microglia activation, and reduced the production of the inflammatory factors tumor necrosis factor-α and interleukin-1β. These results suggest that miR-455-5p is a potential biomarker and therapeutic target for the treatment of cerebral ischemia/reperfusion injury and that it alleviates cerebral ischemia/reperfusion injury by inhibiting C-C chemokine receptor type 5 expression and reducing the neuroinflammatory response.
Related collections
Most cited references46

- Record: found
- Abstract: found
- Article: found
Neuroinflammation: friend and foe for ischemic stroke
- Record: found
- Abstract: found
- Article: not found
Ischaemic stroke
- Record: found
- Abstract: found
- Article: not found