- Record: found
- Abstract: found
- Article: not found
In macrophages, HIV-1 assembles into an intracellular plasma membrane domain containing the tetraspanins CD81, CD9, and CD53
Read this article at
Abstract
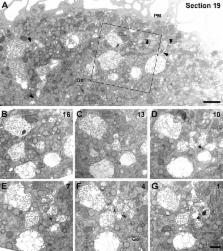
In macrophages, HIV-1 has been shown to bud into intracellular structures that contain the late endosome marker CD63. We show that these organelles are not endosomes, but an internally sequestered plasma membrane domain. Using immunofluorescence microscopy and immunoelectron microscopy, we find that HIV-1 buds into a compartment that contains the tetraspanins CD81, CD9, and CD53. On uninfected macrophages, these proteins are seen at the cell surface and in intracellular vacuole-like structures with a complex content of vesicles and interconnected membranes that lack endosome markers, including CD63. Significantly, these structures are accessible to small tracers (horseradish peroxidase or ruthenium red) applied to cells at 4°C, indicating that they are connected to the cell surface. HIV assembles on, and accumulates within, these intracellular compartments. Furthermore, CD63 is recruited to the virus-containing structures and incorporated into virions. These results indicate that, in macrophages, HIV-1 exploits a previously undescribed intracellular plasma membrane domain to assemble infectious particles.
Related collections
Most cited references40
- Record: found
- Abstract: found
- Article: not found
Selective enrichment of tetraspan proteins on the internal vesicles of multivesicular endosomes and on exosomes secreted by human B-lymphocytes.
- Record: found
- Abstract: found
- Article: not found
Recruitment of HIV and its receptors to dendritic cell-T cell junctions.
- Record: found
- Abstract: found
- Article: not found
Structural basis for targeting HIV-1 Gag proteins to the plasma membrane for virus assembly.
Author and article information
Comments
Comment on this article
Similar content90
Cited by113
- ExosomesAuthors: D Pegtel, Stephen Gould
