- Record: found
- Abstract: found
- Article: found
T Cells That Help B Cells in Chronically Inflamed Tissues
Read this article at
Abstract
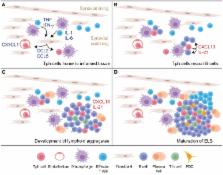
Chronically inflamed tissues commonly accrue lymphocyte aggregates that facilitate local T cell-B cell interactions. These aggregates can range from small, loosely arranged lymphocyte clusters to large, organized ectopic lymphoid structures. In some cases, ectopic lymphoid structures develop germinal centers that house prototypical T follicular helper (Tfh) cells with high expression of Bcl6, CXCR5, PD-1, and ICOS. However, in many chronically inflamed tissues, the T cells that interact with B cells show substantial differences from Tfh cells in their surface phenotypes, migratory capacity, and transcriptional regulation. This review discusses observations from multiple diseases and models in which tissue-infiltrating T cells produce factors associated with B cell help, including IL-21 and the B cell chemoattractant CXCL13, yet vary dramatically in their resemblance to Tfh cells. Particular attention is given to the PD-1 hi CXCR5 − Bcl6 low T peripheral helper (Tph) cell population in rheumatoid arthritis, which infiltrates inflamed synovium through expression of chemokine receptors such as CCR2 and augments synovial B cell responses via CXCL13 and IL-21. The factors that regulate CD4 + T cell production of CXCL13 and IL-21 in these settings are also discussed. Understanding the range of T cell populations that can provide help to B cells within chronically inflamed tissues is essential to recognize these cells in diverse inflammatory conditions and to optimize either broad or selective therapeutic targeting of B cell-helper T cells.
Related collections
Most cited references86
- Record: found
- Abstract: found
- Article: not found
Meningeal B-cell follicles in secondary progressive multiple sclerosis associate with early onset of disease and severe cortical pathology.
- Record: found
- Abstract: found
- Article: not found
Follicular B Helper T Cells Express Cxc Chemokine Receptor 5, Localize to B Cell Follicles, and Support Immunoglobulin Production
- Record: found
- Abstract: found
- Article: not found