- Record: found
- Abstract: found
- Article: found
Prevention, treatment and potential mechanism of herbal medicine for Corona viruses: A review

Read this article at
ABSTRACT
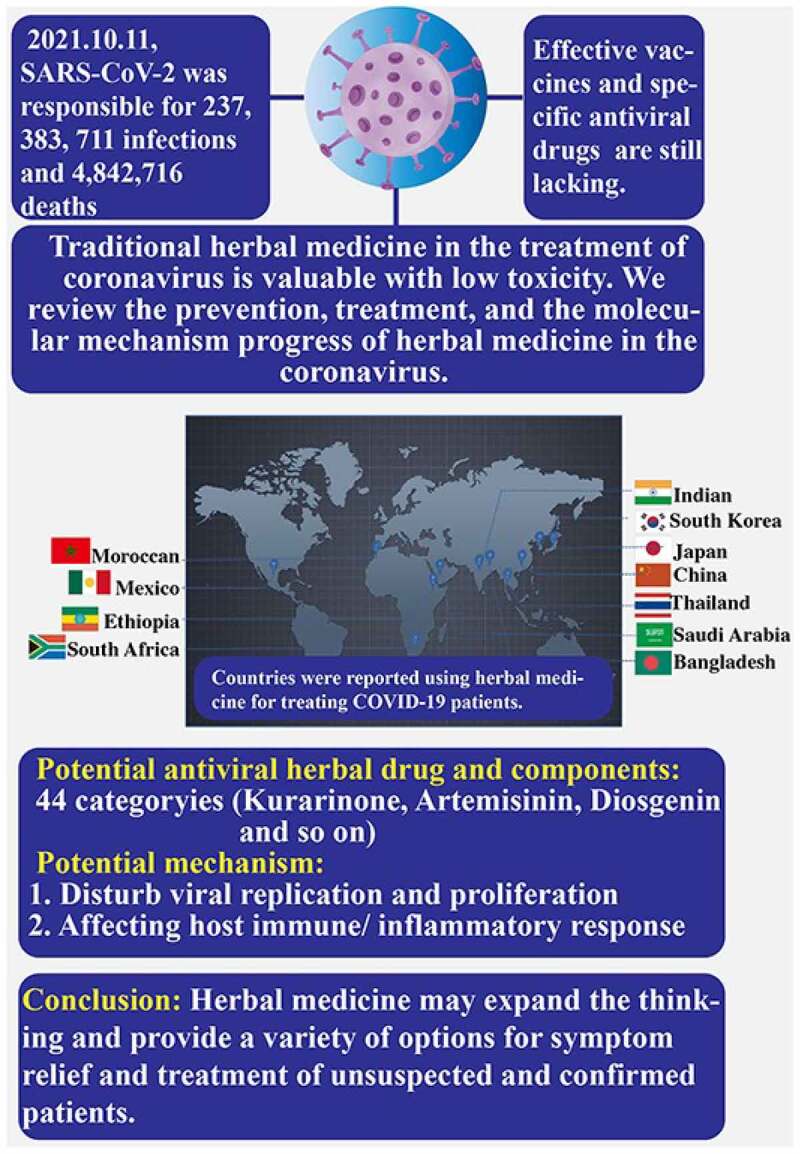
The pandemic of coronavirus disease 2019 (COVID-19) caused by the SARS-coronavirus 2(SARS-CoV-2) virus has become the greatest global public health crisis in recent years,and the COVID-19 epidemic is still continuing. However, due to the lack of effectivetherapeutic drugs, the treatment of corona viruses is facing huge challenges. In thiscontext, countries with a tradition of using herbal medicine such as China have beenwidely using herbal medicine for prevention and nonspecific treatment of corona virusesand achieved good responses. In this review, we will introduce the application of herbalmedicine in the treatment of corona virus patients in China and other countries, andreview the progress of related molecular mechanisms and antiviral activity ingredients ofherbal medicine, in order to provide a reference for herbal medicine in the treatment ofcorona viruses. We found that herbal medicines are used in the prevention and fightagainst COVID-19 in countries on all continents. In China, herbal medicine has beenreported to relieve some of the clinical symptoms of mild patients and shorten the length of hospital stay. However, as most herbal medicines for the clinical treatment of COVID-19still lack rigorous clinical trials, the clinical and economic value of herbal medicines in theprevention and treatment of COVID-19 has not been fully evaluated. Future work basedon large-scale randomized, double-blind clinical trials to evaluate herbal medicines andtheir active ingredients in the treatment of new COVID-19 will be very meaningful.
Graphical abstract
Related collections
Most cited references167
- Record: found
- Abstract: found
- Article: not found
Clinical features of patients infected with 2019 novel coronavirus in Wuhan, China

- Record: found
- Abstract: found
- Article: found
A pneumonia outbreak associated with a new coronavirus of probable bat origin
- Record: found
- Abstract: found
- Article: not found