- Record: found
- Abstract: found
- Article: not found
The Transcription Factor T-bet Regulates Mucosal T Cell Activation in Experimental Colitis and Crohn's Disease
Read this article at
Abstract
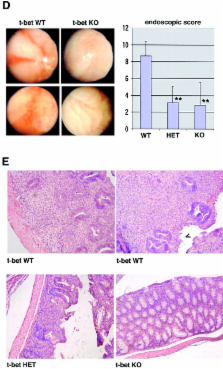
The balance between pro and antiinflammatory cytokines secreted by T cells regulates both the initiation and perpetuation of inflammatory bowel diseases (IBD). In particular, the balance between interferon (IFN)-γ/interleukin (IL)-4 and transforming growth factor (TGF)-β activity controls chronic intestinal inflammation. However, the molecular pathways that evoke these responses are not well understood. Here, we describe a critical role for the transcription factor T-bet in controlling the mucosal cytokine balance and clinical disease. We studied the expression and function of T-bet in patients with IBD and in mucosal T cells in various T helper (Th)1- and Th2-mediated animal models of chronic intestinal inflammation by taking advantage of mice that lack T-bet and retroviral transduction techniques, respectively. Whereas retroviral transduction of T-bet in CD62L + CD4 + T cells exacerbated colitis in reconstituted SCID mice, T-bet–deficient T cells failed to induce colitis in adoptive transfer experiments suggesting that overexpression of T-bet is essential and sufficient to promote Th1-mediated colitis in vivo. Furthermore, T-bet–deficient CD62L − CD4 + T cells showed enhanced protective functions in Th1-mediated colitis and exhibited increased TGF-β signaling suggesting that a T-bet driven pathway of T cell activation controls the intestinal balance between IFN-γ/IL-4 and TGF-β responses and the development of chronic intestinal inflammation in T cell–mediated colitis. Furthermore, TGF-β was found to suppress T-bet expression suggesting a reciprocal relationship between TGF-β and T-bet in mucosal T cells. In summary, our data suggest a key regulatory role of T-bet in the pathogenesis of T cell–mediated colitis. Specific targeting of this pathway may be a promising novel approach for the treatment of patients with Crohn's disease and other autoimmune diseases mediated by Th1 T lymphocytes.
Related collections
Most cited references55
- Record: found
- Abstract: found
- Article: not found
Targeted disruption of the mouse transforming growth factor-beta 1 gene results in multifocal inflammatory disease.
- Record: found
- Abstract: found
- Article: not found
A short-term study of chimeric monoclonal antibody cA2 to tumor necrosis factor alpha for Crohn's disease. Crohn's Disease cA2 Study Group.
- Record: found
- Abstract: found
- Article: not found
