- Record: found
- Abstract: found
- Article: found
Modulation of Opioid Transport at the Blood-Brain Barrier by Altered ATP-Binding Cassette (ABC) Transporter Expression and Activity
Read this article at
Abstract
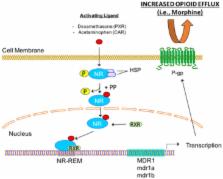
Opioids are highly effective analgesics that have a serious potential for adverse drug reactions and for development of addiction and tolerance. Since the use of opioids has escalated in recent years, it is increasingly important to understand biological mechanisms that can increase the probability of opioid-associated adverse events occurring in patient populations. This is emphasized by the current opioid epidemic in the United States where opioid analgesics are frequently abused and misused. It has been established that the effectiveness of opioids is maximized when these drugs readily access opioid receptors in the central nervous system (CNS). Indeed, opioid delivery to the brain is significantly influenced by the blood-brain barrier (BBB). In particular, ATP-binding cassette (ABC) transporters that are endogenously expressed at the BBB are critical determinants of CNS opioid penetration. In this review, we will discuss current knowledge on the transport of opioid analgesic drugs by ABC transporters at the BBB. We will also examine how expression and trafficking of ABC transporters can be modified by pain and/or opioid pharmacotherapy, a novel mechanism that can promote opioid-associated adverse drug events and development of addiction and tolerance.
Related collections
Most cited references144
- Record: found
- Abstract: found
- Article: not found
Clinical guidelines for the use of chronic opioid therapy in chronic noncancer pain.
- Record: found
- Abstract: found
- Article: not found
Rates of opioid misuse, abuse, and addiction in chronic pain: a systematic review and data synthesis.
- Record: found
- Abstract: found
- Article: not found