- Record: found
- Abstract: found
- Article: found
No ATG8s, no problem? How LC3/GABARAP proteins contribute to autophagy
article-commentary
Read this article at
There is no author summary for this article yet. Authors can add summaries to their articles on ScienceOpen to make them more accessible to a non-specialist audience.
Abstract
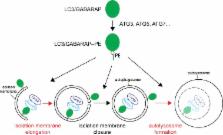
Martens previews work by Nguyen et al. analyzing the essential functions of ATG8 family proteins LC3/GABARAPs in autophagy.
Abstract
The ATG8 family LC3/GABARAP proteins are attached to the membrane of nascent autophagosomes, but their functions during autophagy are unclear. In this issue, Nguyen et al. (2016. J. Cell Biol. https://doi.org/10.1083/jcb.201607039) show that LC3/GABARAP proteins are not essential for autophagosome formation but are critical for autophagosome–lysosome fusion.
Related collections
Most cited references8
- Record: found
- Abstract: found
- Article: not found
A ubiquitin-like system mediates protein lipidation.
Y Ichimura, T Kirisako, T Takao … (2000)
- Record: found
- Abstract: found
- Article: not found
Discovery of Atg5/Atg7-independent alternative macroautophagy.
Yuya Nishida, Satoko Arakawa, Kenji Fujitani … (2009)

- Record: found
- Abstract: found
- Article: found
Atg8 family LC3/GABARAP proteins are crucial for autophagosome–lysosome fusion but not autophagosome formation during PINK1/Parkin mitophagy and starvation
Thanh Nguyen, Benjamin Padman, Joanne Usher … (2016)