- Record: found
- Abstract: found
- Article: found
Sustainable use of CRISPR/Cas in fish aquaculture: the biosafety perspective
Read this article at
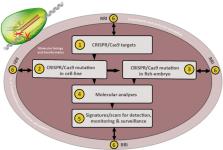
Abstract
Aquaculture is becoming the primary source of seafood for human diets, and farmed fish aquaculture is one of its fastest growing sectors. The industry currently faces several challenges including infectious and parasitic diseases, reduced viability, fertility reduction, slow growth, escapee fish and environmental pollution. The commercialization of the growth-enhanced AquAdvantage salmon and the CRISPR/Cas9-developed tilapia ( Oreochromis niloticus) proffers genetic engineering and genome editing tools, e.g. CRISPR/Cas, as potential solutions to these challenges. Future traits being developed in different fish species include disease resistance, sterility, and enhanced growth. Despite these notable advances, off-target effect and non-clarification of trait-related genes among other technical challenges hinder full realization of CRISPR/Cas potentials in fish breeding. In addition, current regulatory and risk assessment frameworks are not fit-for purpose regarding the challenges of CRISPR/Cas notwithstanding that public and regulatory acceptance are key to commercialization of products of the new technology. In this study, we discuss how CRISPR/Cas can be used to overcome some of these limitations focusing on diseases and environmental release in farmed fish aquaculture. We further present technical limitations, regulatory and risk assessment challenges of the use of CRISPR/Cas, and proffer research strategies that will provide much-needed data for regulatory decisions, risk assessments, increased public awareness and sustainable applications of CRISPR/Cas in fish aquaculture with emphasis on Atlantic salmon ( Salmo salar) breeding.
Related collections
Most cited references103
- Record: found
- Abstract: found
- Article: not found
Repurposing CRISPR as an RNA-guided platform for sequence-specific control of gene expression.
- Record: found
- Abstract: found
- Article: not found
Repair of double-strand breaks induced by CRISPR–Cas9 leads to large deletions and complex rearrangements
- Record: found
- Abstract: found
- Article: found